Identification of Breast Cancer Patients at Risk for Bone Metastasis – A Case–Control Study
Case Report | Volume 5 | Issue 2 | JBST May – August 2019 | Page 15-19| Purnima Thakur, Vishal Verma, Ekta Dogra, Ankur Gupta, Manish Gupta, Vikas Fotedar. DOI: 10.13107/jbst.2019.v05i02.430
Author Purnima Thakur[1], Vishal Verma[2], Ekta Dogra[3], Ankur Gupta[4], Manish Gupta[1], Vikas Fotedar[1]
[1]Department of Radiotherapy, Indira Gandhi Medical College, Shimla, Himachal Pradesh, India,
[2]Department of Orthopedics, Indira Gandhi Medical College, Shimla, Himachal Pradesh, India,
[3]Department of Community Medicine, Postgraduate Institute of Medical Education and Research, Chandigarh, India,
[4]Department of Anatomy, Indira Gandhi Medical College, Shimla, Himachal Pradesh, India.
Address of Correspondence
Dr. Vishal Verma,
Department of Orthopedics, Indira Gandhi Medical College, Shimla, Himachal Pradesh, India.
E-mail: vishhal82@gmail.com
Abstract
Introduction: Prognostic factors for metastatic breast carcinoma are a less studied topic than a prognostic factor for primary breast cancer. Bone is the most frequent site for metastasis in breast cancer patients. Bone metastasis decreases the survival and impacts the quality of life(QoL)in breast cancer patients. Therefore, the study to identify prognostic factors of this specific group of patients appears to be worthy of more detailed study. To the best of our knowledge, this is the first study of its kind in the North Indian population.
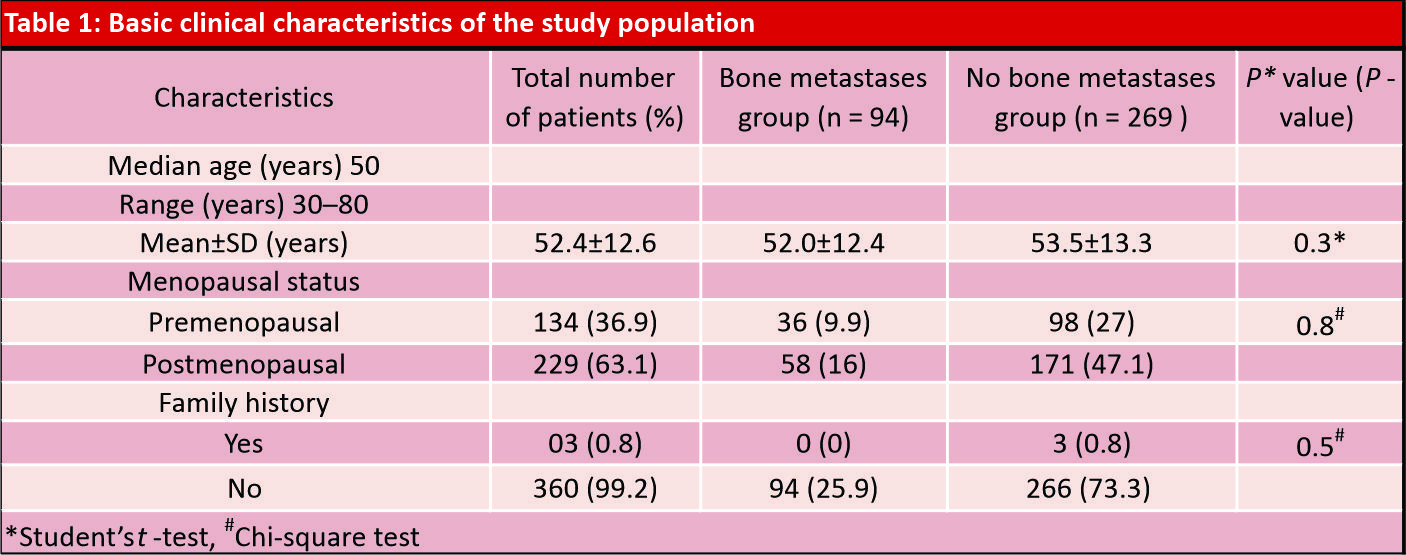
Materials and Methods: A retrospective case–control study was conducted at a tertiary cancer centerin Northern India between January 2011 and December 2015. All patients’ clinical and demographic data were obtained from the medical record of the institute. The incidence and distribution of bone metastases from breast cancer were evaluated, and the correlation between diverse clinical-pathological parameters and bone metastases were analyzed in this study.
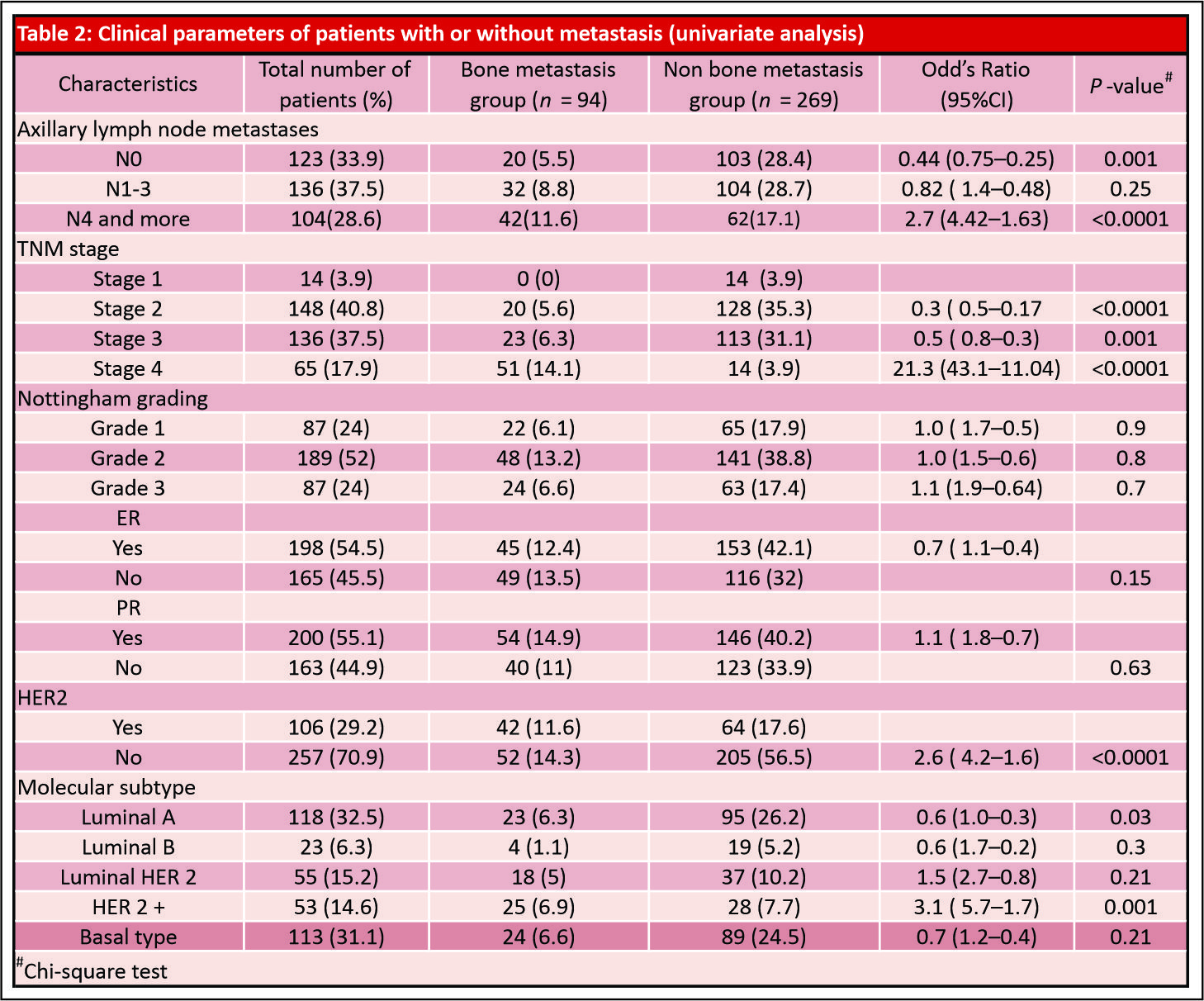
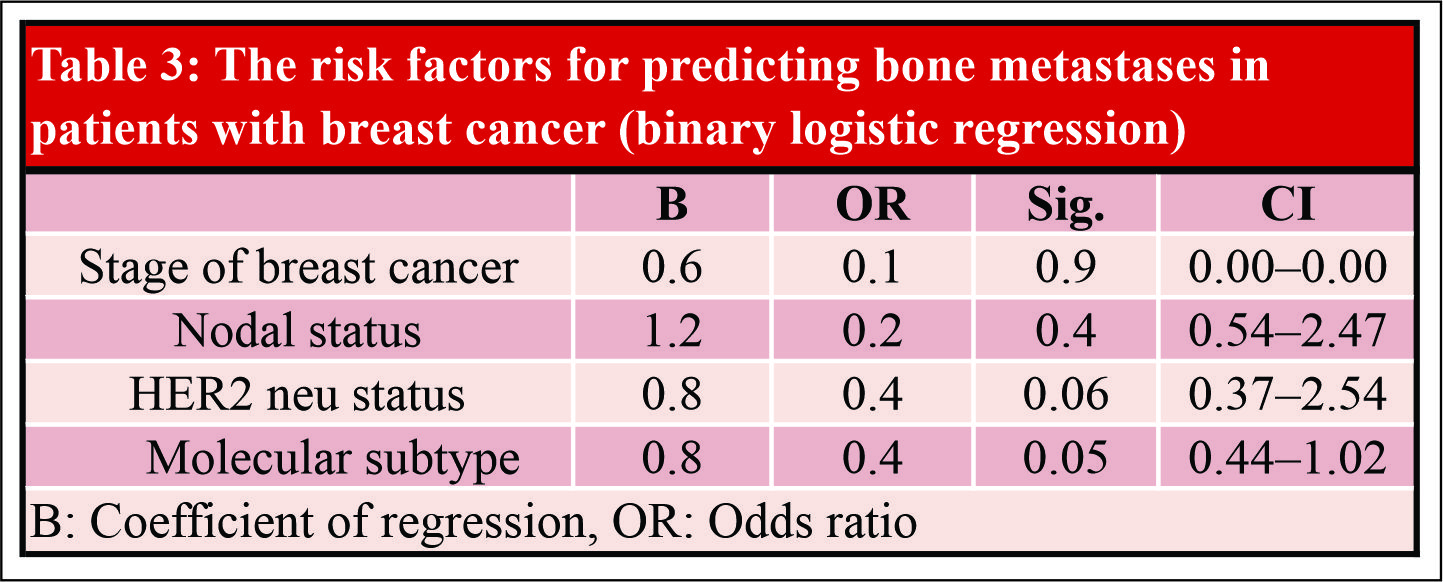
Results: A total of 363 patients were recruited, including 94 cases with bone metastases and 269 controls without bone metastases at presentation. Positive axillary lymph node status, higher stage tumors, HER2 neu-negative disease, and histological subtypes of tumor were found significant prognostic factors in univariate analysis associated with higher rates of bone metastasis. Luminal A(estrogen receptor-positive [ER+], progesterone receptor-positive [PR+], HER2–ve, Grade 1,2), and HER 2 enriched (ER –ve, PR–ve, and HER 2+) molecular subtypes are associated with increased risk of bone metastasis. None of the risk factors studied were significantly associated with bonemetastasis in binary logistic regression analysis.
Conclusion: Identification of breast cancer patients at risk for bone metastasis may aid in the prevention, prediction, detection, and early treatment of these lesions, thus providing improved survival and better QoL.
Keywords: Bone metastasis, risk factors, breast cancer.
References
1. Liede A, Jerzak KJ, Hernandez RK, Wade SW, Sun P, Narod SA, et al. The incidence of bone metastasis after early-stage breast cancer in Canada. Breast Cancer Res Treat 2016;156:587-95.
2. Coleman RE. Metastatic bone disease: Clinical features, pathophysiology and treatment strategies. Cancer Treat Rev 2001;27:165-76.
3. Koizumi M, Yoshimoto M, Kasumi F, Iwase T, Ogata E. Post-operative breast cancer patients diagnosed with skeletal metastasis without bone pain had fewer skeletal-related events and deaths than those with bone pain. BMC Cancer 2010;10:423.
4. Cramarossa G, Chow E, Zhang L, Bedard G, Zeng L, Sahgal A, et al. Predictive factors for overall quality of life in patients with advanced cancer. Support Care Cancer 2013;21:1709-16.
5. Lee SJ, Park S, Ahn HK, Yi JH, Cho EY, Sun JM, et al. Implications of bone-only metastases in breast cancer: Favorable preference with excellent outcomes of hormone receptor positive breast cancer. Cancer Res Treat 2011;43:89-95.
6. Ahn SG, Lee HM, Cho SH, Lee SA, Hwang SH, Jeong J, et al. Prognostic factors for patients with bone-only metastasis in breast cancer. Yonsei Med J 2013;54:1168-77.
7. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D, et al. Global cancer statistics. CA Cancer J Clin 2011;61:69-90.
8. Parkin DM. Cancer base No. 7. In: Cancer Incidence in Five Continents. Vol. 1-8. Lyon: IARC Press; 2005.
9. Brockton NT, Gill SJ, Laborge SL, Paterson AH, Cook LS, Vogel HJ, et al. The breast cancer to bone (B2B) metastases research program: A multi-disciplinary investigation of bone metastases from breast cancer. BMC Cancer 2015;15:512.
10. Pulido C, Vendrell I, Ferreira AR, Casimiro S, Mansinho A, Alho I, et al. Bone metastasis risk factors in breast cancer. Ecancermedicalscience2017;11:715.
11. Chen WZ, Shen JF, Zhou Y, Chen XY, Liu JM, Liu ZL, et al. Clinical characteristics and risk factors for developing bone metastases in patients with breast cancer. Sci Rep 2017;7:11325.
12. Colleoni M, O’Neill A, Goldhirsch A, Gelber RD, Bonetti M, Thürlimann B, et al. Identifying breast cancer patients at high risk for bone metastases. J Clin Oncol 2000;18:3925-35.
13. James JJ, Evans AJ, Pinder SE, Gutteridge E, Cheung KL, Chan S, et al. Bone metastases from breast carcinoma: Histopathological radiological correlations and prognostic features. Br J Cancer 2003;89:660-5.
14. Purushotham A, Shamil E, Cariati M, Agbaje O, Muhidin A, Gillett C, et al. Age at diagnosis and distant metastasis in breast cancera surprising inverse relationship. Eur J Cancer 2014;50:1697-705.
15. Xiao W, Zheng S, Yang A, Zhang X, Zou Y, Tang H, et al. Breast cancer subtypes and the risk of distant metastasis at initial diagnosis: A population-based study. Cancer Manag Res 2018;10:5329-38.
16. Diessner J, Wischnewsky M, Stüber T, Stein R, Krockenberger M, Häusler S, et al. Evaluation of clinical parameters influencing the development of bone metastasis in breast cancer. BMC Cancer 2016;16:307.
17. Irawan C, Hukom R, Prayogo N. Factors associated with bone metastasis in breast cancer: A preliminary study in an Indonesian population. Acta Med Indones2008;40:178-80.
18. Coleman RE, Rubens RD. The clinical course of bone metastases from breast cancer. Br J Cancer 1987;55:61-6.
19. Huszno J, Nowara E. Risk factors for disease progression in HER2-positive breast cancer patients based on the location of metastases. PrzMenopauzalny2015;14:173-7.
20. Yazdani A, Dorri S, Atashi A, Shirafkan H, Zabolinezhad H. Bone metastasis prognostic factors in breast cancer. Breast Cancer (Auckl) 2019;13:1178223419830978.
 |
 |
 |
 |
 |
 |
| Dr. Purnima Thakur | Dr. Vishal Verma | Dr. Ekta Dogra | Dr. Ankur Gupta | Dr. Manish Gupta | Dr. Vikas Fotedar |
| How to Cite this article: Thakur P, Verma V, Dogra E, Gupta A, Gupta M, Fotedar V. Identification of Breast Cancer Patients at Risk for Bone Metastasis – A Case–Control Study. Journal of Bone and Soft Tissue Tumors May-August 2019;5(2): 15-19. |