Journal of Bone and Soft Tissue Tumors (JBST) Editorial Office
For any queries please contact the editorial office
Journal of Bone and Soft Tissue Tumors (JBST)
Ediotrial office, IORG House, A-203, Manthan Apts, Shreesh CHS, Hajuri Road, Thane [W]
Maharashtra, India. Pin 400604
Tel- 02225834545 (office time 10-5 pm, Mon-Fri)
Email: editor.jbst@gmail.com
Publisher: Indian Orthopaedic Research Group. Thane, India. 400604
Email: indian.ortho@gmail.com | Tel: 022-25834545
Important Information
Journal of Bone and Soft Tissue Tumors (JBST) License
![]()
Journal of Bone and Soft Tissue Tumors (JBST) by http://jbstjournal.com/ is licensed under https://creativecommons.org/licenses/by-nc-sa/4.0/
Based on a work at http://jbstjournal.com/.
Permissions beyond the scope of this license may be available at editor.jbst@gmail.com
Journal of Bone and Soft Tissue Tumors (JBST) is the official Journal of The Indian Musculo Skeletal Oncology Society


Limb Salvage With Megaprosthesis In Extremity Osteosarcoma –a Case-based Approach
Volume 2 | Issue 2 | May-Aug 2016 | Page 13-19 | Gurpal Singh, Mark Edward Puhaindran.
Authors: Gurpal Singh[1], Mark Edward Puhaindran[1].
[1]Orthopedic Oncology Services, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Address of Correspondence
Division of Musculoskeletal Oncology, University Orthopaedics, Hand and Reconstructive Microsurgery Cluster, National University Health System, Singapore.
Abstract
Introduction: Musculoskeletal oncology is an evolving field in orthopaedic surgery and surgical management for patients with osteosarcoma has changed fundamentally over the past three decades. Osteosarcoma is a rare tumour, but it is the most common type of primary bone cancer with a biomodal peak. Primary cases tend to occur within the first two decades of life and secondary osteosarcomas affect older patients. Common causes of secondary osteosarcoma include Paget’s disease of bone and radiation exposure. Due to advances in chemotherapeutic regimes, imaging modalities, surgical techniques, material and prosthesis designs, amputation is no longer considered as treatment of choice for most patients. Limb salvage surgery including endoprosthetic reconstructions is also constantly evolving, addressing surgical challenges such as margin control, reconstructive procedures and soft tissue management. In this review, we focus our discussion on the management of patients with osteosarcoma undergoing megaprosthetic reconstruction. Based on three real cases (osteosarcoma of the distal femur, pelvic osteosarcoma involving the hip joint and osteosarcoma of the proximal tibia), this paper aims to highlight surgical challenges that sometimes need to be overcome in this very challenging field.
Keywords: Limb salvage surgery; megaprosthetic reconstruction; osteosarcoma; musculoskeletal oncology.
Introduction
Musculoskeletal oncology is a rapidly evolving field in orthopaedic surgery and surgical management for patients with osteosarcoma has progressed significantly over the past three decades. Due to advances in neoadjuvant treatment, imaging modalities, surgical techniques and material and prosthesis designs, amputation is no longer considered as standard of care in most cases. Literature is currently reporting limb salvage surgeries in 85% to 95% without compromising oncological principles when compared to amputation [1–5]. Main objectives of limb salvage surgery include maximized functional outcome, satisfactory wound coverage for adjuvant therapy and optimized aesthetic outcome without compromising oncologic principles [6]. The reconstructive options for limb salvage surgery can be thought of as biological (autograft, allograft etc.) or endoprosthetic. Sometimes, a combination of both may be necessary. When considering treatment options for patients with osteosarcoma, several factors need to be considered, including the response to neoadjuvant chemotherapy, evidence of periprosthetic fracture, anatomic site, involvement of soft tissue, joint and neurovascular structures as well as the evidence of metastases at diagnosis. Adequate margin control during surgery is crucial. Surgery represents only one component of the multidisciplinary treatment protocol of osteosarcoma patients. Since the 1970s, the role of neoadjuvant therapy has evolved and intense multi-agent chemotherapy has improved the prognosis significantly by eradicating accompanying micrometastases and also reducing the reactive zone around the tumour. The main drawback of chemotherapy prior to surgery includes immunosuppression. Therefore, a multidisciplinary approach between medical oncologists and the surgical team is essential in order to optimise treatment for the patient. After completion of neoadjuvant chemotherapy, re-staging is performed to assess response to chemotherapy and verify resectability and evaluate margins prior to proceeding with definite surgery. Limb salvage surgery for osteosarcoma is typically described as consisting of three parts, starting with en bloc resection of the tumour. Based on the exact location, intra- or extra-articular resection may be considered. In skeletally immature patients, the possibility of physis preservation should be considered. Thereafter, the bone defect is reconstructed. The third part of the surgery consists of soft tissue coverage and functional re-establishment. Treatment protocols for osteosarcoma further include the administration of adjuvant chemotherapy and subsequent long-term surveillance. After limb salvage surgery, overall survival rates of 60% to 70% at 5 years have been reported in literature [1–4, 9, 15]. A recent systematic review and meta-analysis by Stokke et al. evaluating the quality of life among paediatric, adolescent and young adult bone tumour survivors suggested that quality of time improves over time. According to their results, female patients and patients at older age are more likely to have a poor quality of life. Interestingly, there was no difference in outcomes between patients who underwent limb salvage surgery versus amputation for local control [16]. Failure rates in patients undergoing reconstruction with megaprostheses are generally higher compared to patients treated with conventional arthroplasties [7]. However, a direct comparison of study findings is difficult due to different settings and definitions used. Failure rates between 40% to 73% at 5 to 15 years have been reported from factors due to the underlying disease contributing to unfavourable early and late surgical outcome, complications can be specifically related to endoprosthetic reconstruction including aseptic loosening, periprosthetic fracture, infection, implant failure, dissociation of modular components and wear [1, 2, 5, 8, 9]. Implant survival rates for fixed-hinge prostheses have been reported to be approximately 70%, whereas rotating hinge prostheses survival between almost 80 to 100% have been reported [1, 7, 10–13]. A classification system proposed by Henderson et al. for megaprosthesis failure indicates 5 causes of failure, including soft tissue failure (Type 1), aseptic loosening (Type 2), structural fracture (Type 3), infection (Type 4) and local tumour recurrence (Type 5) [14].
Three case examples of patients with osteosarcoma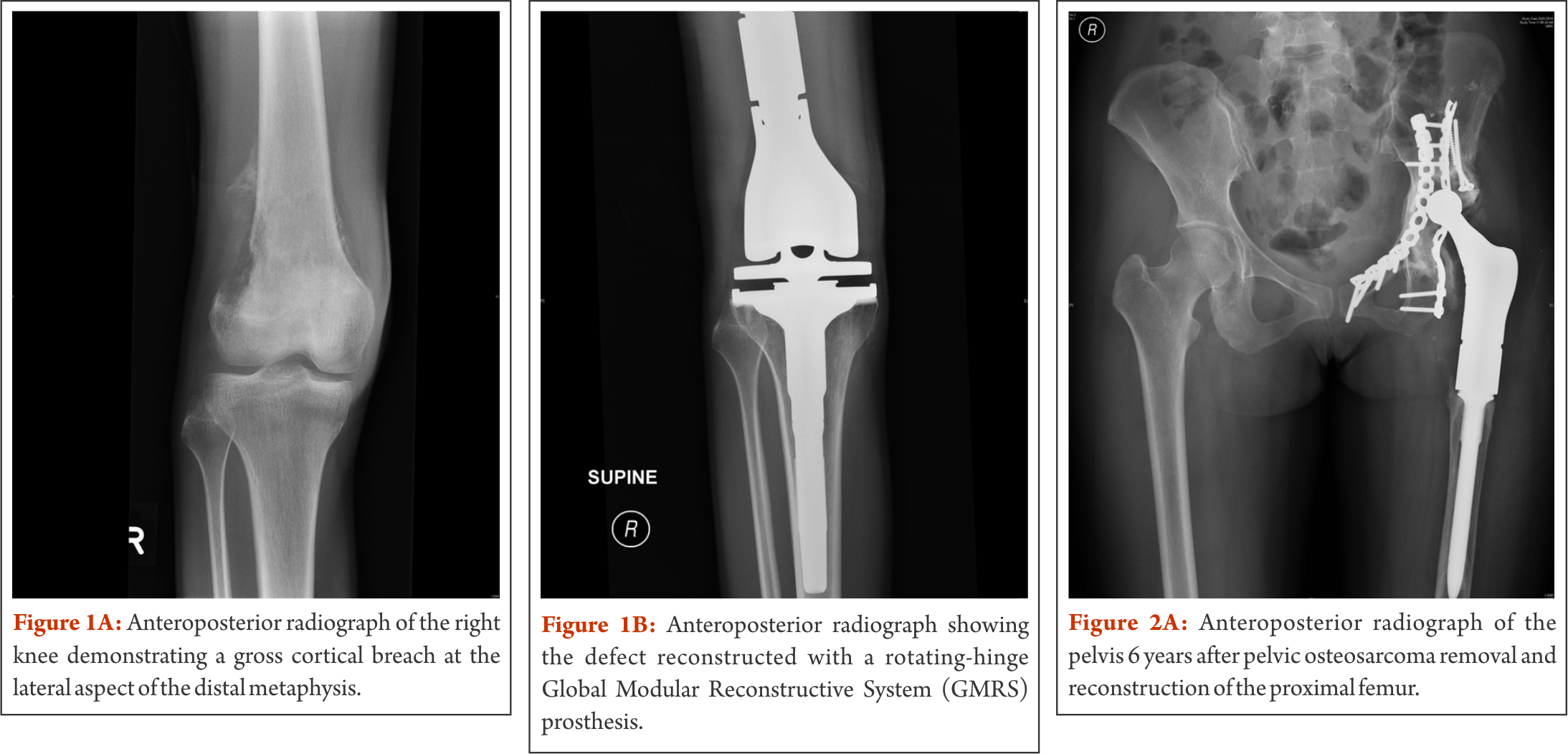
Case 1 – Osteosarcoma of the distal femur
A 16 year old male patient presented with right knee pain at the lateral aspect of the knee and night pain for about one month. There was no known trauma to the right knee. Clinical findings revealed tenderness over the medial and lateral aspect of the knee and a decreased range of motion 10-110°. Plain radiograph, magnetic resonance imaging, computed tomography and bone scan for local and distal staging were performed, showing an aggressive right distal femoral tumour, with extraosseous soft tissue extension abutting the quadriceps and gastrocnemius muscles, iliotibial band, and medial and lateral gutters of the knee joint (Figure 1a). No fracture or neurovascular involvement was detected. There was no evidence of distant osseous or pulmonary metastases. An open biopsy confirmed the diagnosis of a high-grade conventional osteosarcoma.
After completion of neoadjuvant chemotherapy, the patient developed an extensive local fungal infection on the lateral aspect of the thigh, surrounding the biopsy incision site. Systemic antifungal therapy was given for two weeks, directed by a dermatologist and surgery was delayed by two weeks. Given the small window of opportunity between neoadjuvant and adjuvant chemotherapy and to minimise further delay in treatment, the surgical team in the outlined case decided to proceed with tumour resection. In view of the recent tinea corporis with lesions on the lateral aspect of the right thigh, the decision was made to proceed with an anteromedial approach with the excision of the biopsy tract laterally. Local tumour recurrence as a result of spread of malignant cells during biopsy has been reported [17, 18]. Consequently, the biopsy tract is considered as contaminated and conventionally excised during tumour removal. However, more recent studies have shown preliminary evidence for the safety of limb salvage surgery without biopsy tract excision following a diagnostic core needle biopsy and fine-needle aspiration, respectively [19, 20]. In the outlined case, a medial dissection via interval between vastus medialis and rectus femoris was performed for en bloc resection of the tumour. Femoral vessels were identified and traced into the popliteal fossa via adductor canal. The femur osteotomy was made at pre-templated 150 mm from the joint line. The sciatic and peroneal nerve were identified and preserved. Histopathological analysis of frozen sections and tumour specimen showed negative margins. After change of instruments, distal femur reconstruction was continued. Femur and tibia were prepared and a rotating-hinge Global Modular Reconstructive System (GMRS) (Stryker Inc, Rutherford, NJ, USA) knee prosthesis was used for reconstruction (Figure 1b). The vastus medialis was mobilised and brought down to cover the defect medially. The extensor mechanism was reconstructed over the prosthesis. No post-operative complications occurred, all wounds healed satisfactorily. Six months after surgery, the patient is walking without any support. This case highlights the necessity of non-conventional surgical approaches when necessary. Although the main tumour burden was on the lateral aspect of the knee joint, given the location of the previous treated fungal infection, the decision for a medial approach was made. Extra- versus intraarticular resection needs to be discussed in the appropriate context. Preliminary evidence is hinting towards an overestimation of neoplasm seeding in the biopsy tract, especially when the biopsy is performed by the operating surgeon himself. The point to emphasize in this case is the close communication between medical oncologist and the surgeon and balancing the risks of delaying adjuvant chemotherapy versus an “unconventional” surgical approach. This patient underwent an intraarticular resection of the distal femur. In cases where the knee joint is found to be contaminated or being directly invaded by the tumour, an extraarticular resection of the entire joint with en-bloc removal is recommended. Usually, this approach is associated with poorer functional results.
Case 2 – Pelvic osteosarcoma involving the hip joint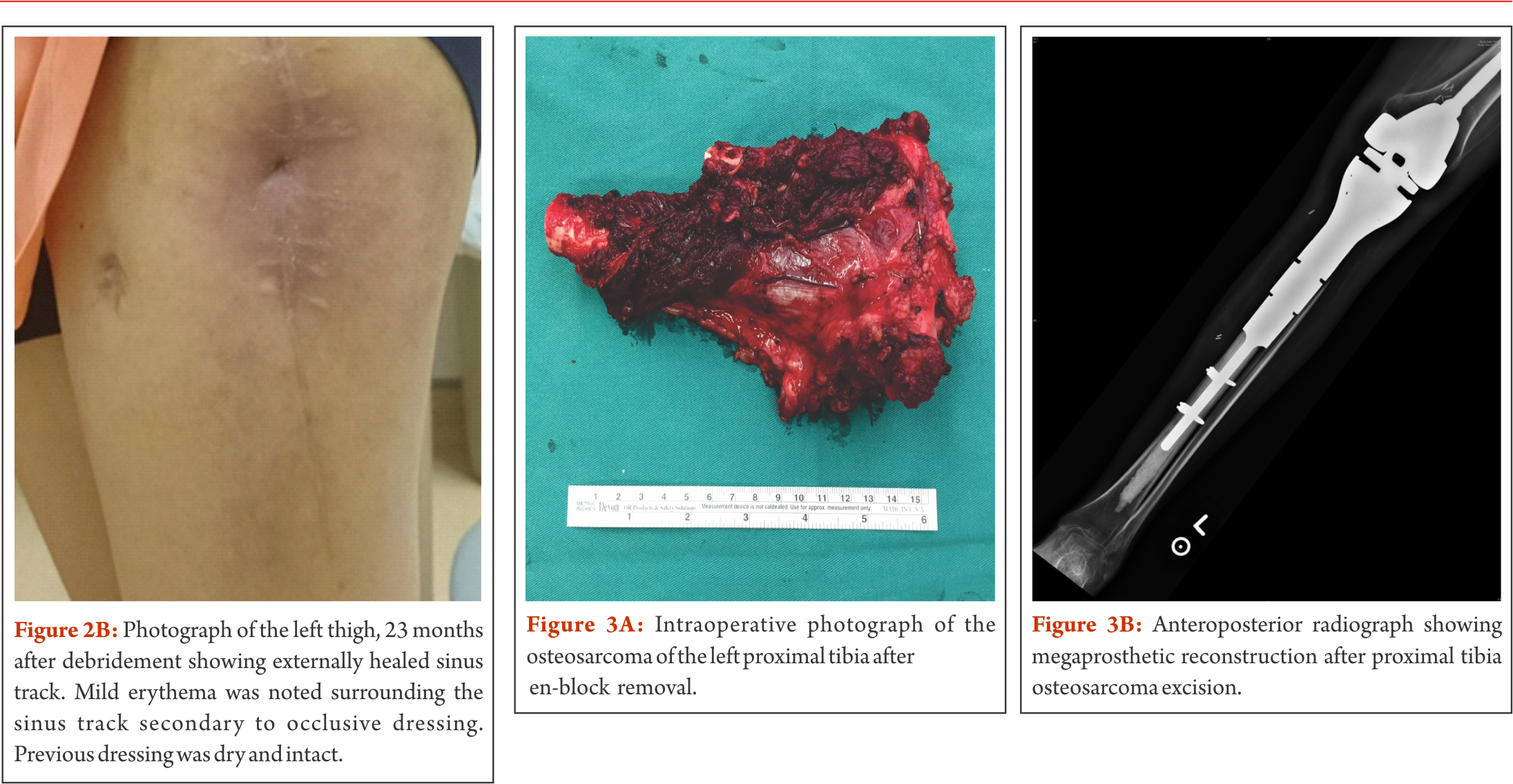
A 24 year old female patient presented with a left neck of femur fracture with underlying osteosarcoma. Prognostic and treatment implications of pathological fractures at presentation have been widely debated. In the past, pathological fractures have been considered as indication for amputation due to the risks of local recurrence from contamination caused by the fracture hematoma. However, a recent systematic review and meta-analysis by Salunke et al. is demonstrating similar rates of local recurrence in patients with limb salvage surgeries compared to amputation, provided carefully selection of patients has been performed [21]. Our own 18-year experience of high-grade osteosarcoma with a pathological fracture at initial presentation showed no differences in the survival and recurrence rates of patients with pathological fractures compared to those with no fractures. Moreover, we did not see any difference in survival between amputated and salvaged patients with fractures, provided margin control attempts are aggressive (unpublished data). In the outlined case, the patient was treated according to protocol with neoadjuvant chemotherapy. Given the complexity of the case, but still aiming for limb salvage, the decision was made for using endoprosthetic and biologic reconstructive options, namely the combination of a hemipelvectomy with an extra-articular proximal femur resection followed by proximal femur replacement and acetabular column plating/deep frozen bone (Fig. 2a). Biologic reconstruction options such as allografts, autografts and re-implantation of sterilized tumour bone replace the defect with a biologic construct, providing the theoretical advantage of incorporation of the biological graft. However, retrieval studies have shown that grafts become necrotic and may act as a spacer rather than a stable replacement. Typical failures include graft-fracture, non-union between the graft and host-bone and increased infection-rates. Drawbacks also include the dependence of graft incorporation for rehabilitation. A reliable tissue bank and associated logistics are sine qua non, and harvesting the autograft or preparing the allograft and subsequently increasing surgical time need to be taken into account. Since a long-term study showed that less than 50% of allografts lasted less than 10 years [22], this technique is mostly preserved for special occasions only, such as osteosarcomas in skeletally immature patients without involving the growth plates. Combined usage of endoprosthetic and biologic reconstruction is described by the allograft prosthetic composite. The concept behind includes restoration of the bone stock by allograft and using endoprosthetic components to restore articulating surfaces, theoretically offering a higher durability in the long-term when compared to pure biologic reconstruction. In the outlined case, the patient’s own pre-treated tumour bone was used for part of the recontruction. Sterilization of the bone can be performed in various ways including pasteurizing, autoclaving, radiotherapy, and freezing. Similarly to the use of allografts, non-union, infection and fracture belong to the main complications in biologic reconstruction with autografts. As described in the outlined case, liquid nitrogen was used for pre-treatment of the bone. Cryosurgery in combination with autograft-prosthesis composites shows promising results with excellent functional outcomes, low complication rates and improved union rates, especially when used with the pedicle-freezing technique [23–25]. After completion of adjuvant chemotherapy and being in remission, the patient in the outlined case developed a hematogenous methicillin susceptible staphylocococcus aureus (MSSA) infection of the endoprosthesis six years postoperatively. Infection is still the most common complication in megaprosthetic reconstruction and in general, it is more likely to happen when adequate wound coverage is missing. In primary megaprosthesis infection rates between 2% to 20% have been reported, with the numbers increasing to more than 40% in revision cases [1, 2, 5, 10, 14, 26–31]. The classic approach in periprosthetic infection is a two-stage exchange revision surgery, including the removal of original endoprosthetic components, thorough debridement, administration of intravenous antibiotics, and implantation of a spacer followed by the re-implantation of implants. In the outlined case, this approach was not practical. A consensus with the multidisciplinary musculoskeletal oncology team and the patient was made for management with ultrasound-guided drainage of the left hip joint and suppressive antibiotics with follow-ups by our colleagues for infectious disease. The patient was discharged with peripherally inserted central catheter line in situ. She continued to develop a collection in the left hip and anterior thigh. A formal arthrotomy with extensive debridement and removal of all necrotic tissue was performed. A large bore chest drain was left in situ, exited through quadriceps anteriorly, plus two additional drains subcutaneously. An urostomy pouch was applied and the patient was kept on long term drainage, allowing the sinus track to the left lateral thigh to mature. In the following, kept with suppressive antibiotics (ciprofloxacin and rifampicin), the patient was under close follow-up with a regular change of the urostomy bag. Erythema and tenderness regressed and drainage successively decreased (Figure 2b). The chest drain was removed two months after surgery. On examination, 29 months after debridement the left thigh is non-tender and showed no erythema. Scars to the left tight (sinus track) are well healed and she had no fever, chills or rigor and denied any pain. She is walking with a slight limp and was returned to work recently. She remains on long-term suppressive antibiotics as guided by the infectious disease physician. This case highlights how limb salvage surgery has pushed the boundaries over the last decades. Combining different reconstructive options may be considered for complex cases. This case also illustrates the option of a relatively conservative management strategy for periprosthetic infection in a compliant patient, achieving a satisfactory result.
Case 3 – Osteosarcoma of the proximal tibia
A 16 year old female patient was diagnosed with non metastatic osteosarcoma of the left proximal tibia (stage IIb). She underwent left tibial osteosarcoma resection, reconstruction with megaprosthesis, extensor mechanism reconstruction, medial gastrocnemius flap and split skin graft (Figure 3a and 3b). Proximal tibia resection presents unique local anatomical features which need to be addressed during limb salvage surgery. Neurovascular structures in the popliteal fossa as well as the peroneal nerve between the biceps femoris and the lateral head of the gastrocnemius muscle need to be preserved as much as possible. Displacement of these structures due to a large tumour mass are likely, therefore careful preparation in this area is required. In most cases, biological borders such as perivascular fat or the popliteus muscle are separating the tumour from the neurovascular bundle. Intraoperatively, overstretching these structures due to overdistraction of the femur and tibia (after resecting the tumour) needs to be avoided in order to prevent neuropraxia and endothelial injury. Involvement of tissue adjacent to the tibiofibular joint is usually of concern and this joint is typically included in the resection. Once the patellar tendon has been divided, reconstruction of the extensor mechanism is obligatory. Various techniques to attach the tendon to the endoprostetic implant have been described in a systematic review by Ek et al. including direct fixation using screw/washer or sutures, synthetic soft tissue augments such as tubes, sutures, cerclage wires and non-absorbable tapes and biological augmentation such as graft from the biceps or satorius tendons or gastrocnemius flaps with or without synthetic materials [32]. The authors report a trend towards improved outcomes with biologic reconstructive options. However, there is no clear evidence for a single technique. Extensor lag is of major concern in all surgeries requiring reconstruction of the extensor mechanism. Therefore, postoperative management in the outlined case included immobilization with a long-leg cast and a long-leg brace respectively for six weeks to allow healing. Obtaining full extension and allowing the extensor mechanism to heal was given priority compared to knee flexion in view of its impact on ambulation and there was a strong emphasis on extensor mechanism strengthening during rehabilitation. The close proximity of the proximal tibia to the skin makes the wound more susceptible to infectious complications. Keeping thick flaps during preparation at the beginning of the surgery minimize necrosis of the skin. Typically, a primary medial gastrocnemius flap is used to improve wound coverage with the advantage of offering a larger muscular material and also bypassing a longer distance when compared to the lateral gastrocnemius [33, 34]. This technique has been first described by Dubousset et al. [35]. Skin grafting provides an additional supportive procedure while relieving tension during skin closure and therefore preventing skin flap necrosis. Currently, split-thickness skin graft is considered as gold standard in any major skin loss. Typically, the graft is meshed in order for expansion while keeping the morbidity to the donor side as low as possible. Postoperative management in patients with skin grafts includes a close observation of the donor side for scarring, pain and signs suggestive of infection. This case illustrates a conventional proximal tibia resection, highlighting surgical considerations with regards to anatomical characteristics and postoperative management.
conclusions
Medical management, surgical techniques and prosthetic design have improved significantly during the last three decades. Amputation is no longer considered as standard of care in patients with osteosarcoma in most cases. However, limb salvage with megaprosthesis remains challenging. Complication rates are high, with infection occurring most commonly. A good interaction within a multidisciplinary team in a preferably high-volume centre is required for optimal management. Given the unique features of these patients, “unconventional” approaches and combinations of reconstructive options may be considered sometimes, provided oncologic principles are not compromised.
References
1.Pala E, Trovarelli G, Calabrò T, Angelini A, Abati CN, Ruggieri P. Survival of modern knee tumor megaprostheses: failures, functional results, and a comparative statistical analysis. Clin Orthop Relat Res 2015;473:891–9.
2.Zeegen EN, Aponte-Tinao LA, Hornicek FJ, Gebhardt MC, Mankin HJ. Survivorship analysis of 141 modular metallic endoprostheses at early followup. Clin Orthop Relat Res 2004:239–50.
3.Bacci G, Ferrari S, Bertoni F, Ruggieri P, Picci P, Longhi A, et al. Long-term outcome for patients with nonmetastatic osteosarcoma of the extremity treated at the istituto ortopedico rizzoli according to the istituto ortopedico rizzoli/osteosarcoma-2 protocol: an updated report. J Clin Oncol 2000;18:4016–27.
4.Bacci G, Picci P, Ferrari S, Ruggieri P, Casadei R, Tienghi A, et al. Primary chemotherapy and delayed surgery for nonmetastatic osteosarcoma of the extremities. Results in 164 patients preoperatively treated with high doses of methotrexate followed by cisplatin and doxorubicin. Cancer 1993;72:3227–38.
5.Gosheger G, Gebert C, Ahrens H, Streitbuerger A, Winkelmann W, Hardes J. Endoprosthetic reconstruction in 250 patients with sarcoma. Clin Orthop Relat Res 2006;450:164–71.
6.Heller L, Kronowitz SJ. Lower extremity reconstruction. J Surg Oncol 2006;94:479–89. doi:10.1002/jso.20485.
7.Heisel C, Kinkel S, Bernd L, Ewerbeck V. Megaprostheses for the treatment of malignant bone tumours of the lower limbs. Int Orthop 2006;30:452–7.
8.Mavrogenis AF, Pala E, Angelini A, Ferraro A, Ruggieri P. Proximal tibial resections and reconstructions: clinical outcome of 225 patients. J Surg Oncol 2013;107:335–42.
9.Orlic D, Smerdelj M, Kolundzic R, Bergovec M. Lower limb salvage surgery: modular endoprosthesis in bone tumour treatment. Int Orthop 2006;30:458–64.
10.Ahlmann ER, Menendez LR, Kermani C, Gotha H. Survivorship and clinical outcome of modular endoprosthetic reconstruction for neoplastic disease of the lower limb. J Bone Joint Surg Br 2006;88:790–5.
11.Bhangu AA, Kramer MJ, Grimer RJ, O’Donnell RJ. Early distal femoral endoprosthetic survival: cemented stems versus the Compress implant. Int Orthop 2006;30:465–72.
12.Myers GJC, Abudu AT, Carter SR, Tillman RM, Grimer RJ. Endoprosthetic replacement of the distal femur for bone tumours: long-term results. J Bone Joint Surg Br 2007;89:521–6. doi:10.1302/0301-620X.89B4.18631.
13.Ruggieri P, Mavrogenis AF, Pala E, Abdel-Mota’al M, Mercuri M. Long term results of fixed-hinge megaprostheses in limb salvage for malignancy. Knee 2012;19:543–9.
14.Henderson ER, Groundland JS, Pala E, Dennis JA, Wooten R, Cheong D, et al. Failure mode classification for tumor endoprostheses: retrospective review of five institutions and a literature review. J Bone Joint Surg Am 2011;93:418–29.
15.Eckardt JJ, Eilber FR, Dorey FJ, Mirra JM. The UCLA experience in limb salvage surgery for malignant tumors. Orthopedics 1985;8:612–21.
16.Stokke J, Sung L, Gupta A, Lindberg A, Rosenberg AR. Systematic review and meta-analysis of objective and subjective quality of life among pediatric, adolescent, and young adult bone tumor survivors. Pediatr Blood Cancer 2015;62:1616–29.
17.Schwartz HS, Spengler DM. Needle tract recurrences after closed biopsy for sarcoma: three cases and review of the literature. Ann Surg Oncol 1997;4:228–36.
18.Davies NM, Livesley PJ, Cannon SR. Recurrence of an osteosarcoma in a needle biopsy track. J Bone Joint Surg Br 1993;75:977–8.
19.Saghieh S, Masrouha KZ, Musallam KM, Mahfouz R, Abboud M, Khoury NJ, Haidar R. The risk of local recurrence along the core-needle biopsy tract in patients with bone sarcomas. Iowa Orthop J 2010;30:80–3.
20.Kaffenberger BH, Wakely PE, Mayerson JL. Local recurrence rate of fine-needle aspiration biopsy in primary high-grade sarcomas. J Surg Oncol 2010;101:618–21.
21.Salunke AA, Chen Y, Tan JH, Chen X, Khin LW, Puhaindran ME. Does a pathological fracture affect the prognosis in patients with osteosarcoma of the extremities?: A systematic review and meta-analysis. Bone Joint J 2014;96-B:1396–403. doi:10.1302/0301-620X.96B10.34370.
22.Brigman BE, Hornicek FJ, Gebhardt MC, Mankin HJ. Allografts about the Knee in Young Patients with High-Grade Sarcoma. Clin Orthop Relat Res 2004:232–9.
23.Subhadrabandhu S, Takeuchi A, Yamamoto N, Shirai T, Nishida H, Hayashi K, et al. Frozen Autograft-Prosthesis Composite Reconstruction in Malignant Bone Tumors. Orthopedics 2015;38:e911-8.
24.Jeon D, Kim MS, Cho WH, Song WS, Lee S. Pasteurized autograft-prosthesis composite for distal femoral osteosarcoma. J Orthop Sci 2007;12:542–9. doi:10.1007/s00776-007-1173-7.
25.Jeon D, Kim MS, Cho WH, Song WS, Lee S. Pasteurized autograft-prosthesis composite for reconstruction of proximal tibia in 13 sarcoma patients. J Surg Oncol 2007;96:590–7.
26.Hardes J, Gebert C, Schwappach A, Ahrens H, Streitburger A, Winkelmann W, Gosheger G. Characteristics and outcome of infections associated with tumor endoprostheses. Arch Orthop Trauma Surg 2006;126:289–96.
27.Lee SH, Oh JH, Lee KS, Yoo KH, Kim HS. Infection after prosthetic reconstruction in limb salvage surgery. Int Orthop 2002;26:179–84. doi:10.1007/s00264-001-0328-y.
28.Unwin PS, Cannon SR, Grimer RJ, Kemp HB, Sneath RS, Walker PS. Aseptic loosening in cemented custom-made prosthetic replacements for bone tumours of the lower limb. J Bone Joint Surg Br 1996;78:5–13.
29.Grimer RJ, Belthur M, Chandrasekar C, Carter SR, Tillman RM. Two-stage revision for infected endoprostheses used in tumor surgery. Clin Orthop Relat Res 2002:193–203.
30.Horowitz SM, Lane JM, Otis JC, Healey JH. Prosthetic arthroplasty of the knee after resection of a sarcoma in the proximal end of the tibia. A report of sixteen cases. J Bone Joint Surg Am 1991;73:286–93.
31.Jeys LM, Grimer RJ, Carter SR, Tillman RM. Periprosthetic infection in patients treated for an orthopaedic oncological condition. J Bone Joint Surg Am 2005;87:842–9. doi:10.2106/JBJS.C.01222.
32.Ek EW, Rozen WM, Ek ET, Rudiger HA. Surgical options for reconstruction of the extensor mechanism of the knee after limb-sparing sarcoma surgery: an evidence-based review. Arch Orthop Trauma Surg 2011;131:487–95.
33.McCraw JB, Fishman JH, Sharzer LA. The versatile gastrocnemius myocutaneous flap. Plast Reconstr Surg 1978;62:15–23.
34.Meller I, Ariche A, Sagi A. The role of the gastrocnemius muscle flap in limb-sparing surgery for bone sarcomas of the distal femur: a proposed classification of muscle transfers. Plast Reconstr Surg 1997;99:751–6.
35.Dubousset J, Missenard G, Genin J. Traitement chirurgical conservateur des sarcomes ostéogéniques des membres. Techniques et résultats fonctionnels. Rev Chir Orthop Reparatrice Appar Mot 1985;71:435–50.
(Abstract Full Text HTML) (Download PDF)
Like this:
Is Limb Salvage Surgery a Contra Indication in Pathological Fractures Secondary to Osteosarcoma? Do We Know The Answer?
Volume 2 | Issue 2 | May-Aug 2016 | Page 10-12 | Zeeshan Khan, Shakir Hussain, Simon Carter.
Authors: Zeeshan Khan [1], Shakir Hussain [1], Simon Carter[1].
1Orthopedic Oncology Services, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Address of Correspondence
Dr. Ashish Gulia
Associate Professor, Orthopedic oncology, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Email: aashishgulia@gmail.com
Abstract
Osteosarcoma is the commonest primary bone tumour with a bimodal age distribution. The survivorship of patients with osteosarcoma has improved with advances in chemotherapy making limb salvage surgery the commonest surgical procedure. Pathological fractures associated with osteosarcoma, however are rare and suggests the aggressiveness of the tumour. These patients are considered as a special group due to the variable outcomes reported in the literature due to some special characteristics, prompting the discussion between limb salvage surgery versus ablative surgery.
This article reviews the reasons why this group of patients are considered challenging and also the various outcomes reported in the literature.
Keywords: Pathological fracture, osteosarcoma, outcomes
Introduction
Primary bone and soft tissue sarcomas are rare tumours. Osteosarcoma is the commonest primary bone tumour with a bimodal age distribution and with a reported incidence of 2-3 per million population per year [1, 2]. With advances in chemotherapy, the survivorship of patients with osteosarcoma has improved significantly with various studies revealing similar results with limb salvage surgery when compared with amputation [5]. Contra indications to limb salvage surgery may include involvement of the neurovascular bundle, joint involvement, progression of disease whilst on treatment, patient choice, infection and a pathological fracture (Fig. 1).
A pathological fractureassociated with osteosarcoma at presentation or during treatment is even rarer with a reported incidence of 5-10% [3, 4]. A pathological fracture can be the mode of presentation for osteosarcomas in certain cases whereas it can occur during treatment in others. This is generally considered to be an aggressive biological behaviour of the disease which in turn, historically, has been considered as a poor prognostic factor in the outcome of this special group of patients [6].
Why is this group of patients special?
This select group of patients poses a challenge to the treating orthopaedic surgeon about the modality of surgical procedure. There has been a debate over the years whether these patients should have ablative or limb salvage surgery and if there is a difference in the outcome of both with varying results reported by different authors [8].
What makes these patients special is the associated hematoma with the pathological fracture which is considered to have tumour cells which spreads locally in the tissues [7]. The extent of spread also depends on the anatomic location of fracture and whether it is intra or extra capsular. The disruption of local microvasculature is also considered to be a risk factor for development of metastasis [7]. Understandably, extra articular resection for intra articular extension of tumours is a more challenging procedure particularly when limb salvage surgery is attempted with the reported outcomes of extra articular resections in limb salvage surgery considered to be compromised as well [9]. The local contamination of soft tissues with the tumour cells is also considered to be a risk factor for local recurrence. This prompted the thought that early and aggressive surgery in the form of ablative surgery will halt the progression and spread of disease any further. The presence of a pathological fracture in osteosarcoma, therefore, has been considered as a poor prognostic factor by some authors but not by all [8, 10, 11].
Initial treatment& work up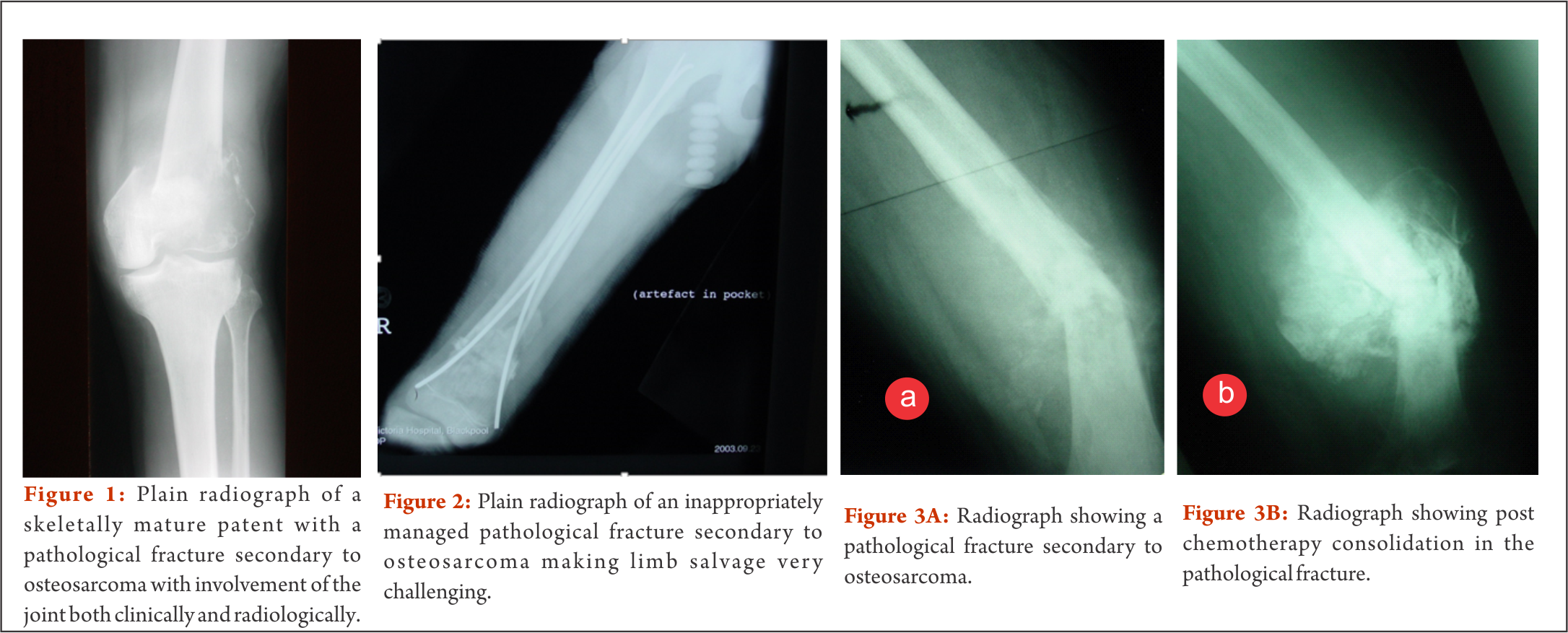
Perhaps the most important step in the management of this select group of patients is the early recognition of the aggressiveness of the lesion and prompt referral to a specialist unit. Failure of recognition of these fractures as being pathological can lead to inappropriate treatment and potentially worse outcomes (Fig. 2) [14]. The rest of the management in a multidisciplinary team setting involves a detailed history, examination of the involved limb and joints for any effusion, local and systemic staging, biopsy and neoadjuvant chemotherapy after confirmation of diagnosis of osteosarcoma.
It is also important to note that one of the most challenging issues with this group of patients is pain management and immobilisation during the pre-operative period whilst they wait for surgery and have neoadjuvant chemotherapy.
Immobilisation
Immobilisation for pain relief can be challenging as this depends on the location of the fracture and may involve a plaster cast, simple sling, skin traction or in some cases external fixators [15]. Significant attention should be paid to the placement of the schanz pins if an external fixator is used due to the risk of tumour spread into non-involved compartments and risk of infection which would compromise limb sparing surgery.
Prognostic factors
A pathological fracture is independently considered a poor prognostic factor in osteosarcoma but was not considered one in cases of chondrosarcoma and Ewing’s sarcoma [8, 10]. A poor response to chemotherapy and local recurrence are also considered to be poor prognostic factors for survivorship [8, 10-13]. It is however, important to note that the efficacy of chemotherapy and healing of fracturesin these special cases are considered as supportive factors for limb salvage surgery [20].
Fracture consolidation
It has been noted that these fractures heal whilst patients are on chemotherapy and in most of the cases these patients have had significant post chemotherapy necrosis(Fig. 3) [8]. On the contrary some fractures might happen whilst patients are on chemo which depicts the aggressive nature of the disease.
What is the verdict?
Limb salvage surgery should be attempted, if possible, in these patients after neoadjuvant treatment but if clear surgical margins cannot be obtained during surgery or limb salvage will result in a poor functioning limb, then ablative surgery should be considered, particularly in the paediatric population where they can adapt to prosthetics earlier than adults [16]. It is however, also important to note that after wide resection of tumour, limb salvage is still a viable option with reconstruction performed with either arthrodesis or rotationplasty where appropriate.
Scully et al, suggested that a pathological was a poor prognostic factor but it is important to note that this study was performed over a 30 year period where some patients in their series had not received any chemortherapy and there have been advances in this field over the study time period [8]. Similarly Finn et al, suggested early amputation due to the risk of local and distant tumour spread [14]. In another study, the 5 year survival in patients with pathological fractures secondary to osteosarcoma was lower than those without a fracture [18]. On the contrary Bacci et al, and Abudu et al, showed that there was no difference in the survivorship of these patients when they were treated with neoadjuvant chemotherapy [7, 11]. In a recent meta-analysis comparing limb salvage with ablative surgery for pathological fractures in high grade osteosarcomas, no significant difference between local recurrence and 5 year survival was noted [19]. Adjuvant radiotherapy in these patients has not been shown to reduce the risk of local recurrence and in fact might increase the risk of these patients undergoing further surgical procedures compromising there outcomes [7].
Future direction
All the studies performed on this select group of patients are retrospective and contain a small number of patients over a prolonged period of time. The results are further effected by variables including the heterogeneity of the patient and fracture characteristics and also the advances in chemotherapy over a period of time. Improvements in surgical techniques have also resulted in improved outcomes. Most of these variables are un avoidable due to the rarity of these cases but in order to come to a definite conclusion, a multi central randomised trial will eradicate all these bias and should guide treatment.
References
1. 1.Bielack S, Carrle D, Jost L. ESMO guidelines working group osteosarcoma: ESMO clinical recommendations for diagnosis, treatment and follow up. Annals of Oncology.2008; 19, supplement 2: 94-96.
2. Widhe B, Widhe T. Initial symptoms and clinical features in osteosarcoma and Ewing sarcoma. J Bone Joint Surg Am. 2000; 82:667-74.
3. Jaffe N, Spears R, Eftekhari F, Robertson R, Cangir A, Takaue Y, Carrasco H, Wallace S, Ayala A,Raymond K, et al. Pathologic fracture in osteosarcoma. Impact of chemotherapy on primarytumorand survival. Cancer. 1987; 59:701-09.
4. Mulder JO, Schutte HE, Kroon HM, Taconis WK. Radiologic atlas of bone tumors. Amsterdam:Elsevier Science. 1993. Intraosseous osteosarcoma: conventional type: 51-5.
5. Simon MA. Current concepts review. Limb salvage in osteosarcoma. J Bone Joint Surg Am.1988; 70:307-10.
6. Coley BL, Pool JL. Factors influencing the prognosis in osteogenic sarcoma. Ann Surg. 1940; 112:1114-28.
7. Abudu A, Sferopoulos NK, Tillman RM, Carter SR, Grimer RJ. The surgical treatment and outcome of pathological fractures in localised osteosarcoma. J Bone Joint Surg Br. 1996; 78:694-8.
8. Scully SP1, Ghert MA, Zurakowski D, Thompson RC, Gebhardt MC. Pathologic fracture in osteosarcoma: prognostic importance and treatment implications. J Bone Joint Surg Am. 2002 Jan;84-A (1):49-57.
9. Hardes J1, Henrichs MP, Gosheger G, Gebert C, Höll S, Dieckmann R, Hauschild G, Streitbürger A. Endoprosthetic replacement after extra-articular resection of bone and soft-tissue tumours around the knee. Bone Joint J. 2013 Oct; 95-B (10):1425-31.
10. Bramer JAM, Abudu AA, Grimer RJ, Carter SR, Tillman RM. Do pathological fractures influence survival and local recurrence rate in bony sarcomas?. Eur J Cancer. 2007 Sep; 43(13):1944-51.
11. Bacci G1, Ferrari S, Longhi A, Donati D, Manfrini M, Giacomini S, Briccoli A, Forni C, Galletti S.Nonmetastatic osteosarcoma of the extremity with pathologic fracture at presentation: local andsystemic control by amputation or limb salvage after preoperative chemotherapy. ActaOrthop Scand. 2003 Aug; 74(4):449-54.
12. Meyers PA, Heller G, Healey J, Huvos A, Lane J, Marcove R, ApplewhiteA, Vlamis V, Rosen G. Chemotherapy for nonmetastatic osteogenic sarcoma: the Memorial Sloan-Kettering experience. J ClinOncol. 1992; 10:5-15.
13. Glasser DB, Lane JM, Huvos AG, Marcove RC, Rosen G. Survival, prognosis, and therapeutic response in osteogenic sarcoma. The Memorial Hospital experience. Cancer. 1992; 69:698-708.
14. Mankin HJ, Mankin CJ, Simon MA. The hazards of the biopsy, revisited: Members of the Musculoskeletal Tumor Society. J Bone Joint Surg [Am] 1996; 78-A: 656–663.
15. Chandrasekar CR, Grimer RJ, Carter SR, et al. Pathological fracture of the proximal femur in osteosarcoma: need for early radical surgery? ISRN Oncol 2012; 2012:512389.
16. Hosalkar HS, Dormans JP. Limb sparing surgery for pediatric musculoskeletal tumors. Pediatr Blood Cancer 2004; 42:295–310.
17. Finn HA, Simon MA. Limb-salvage surgery in the treatment of osteosarcoma in skeletally immature individuals. ClinOrthopRelat Res 1991; 262:108–118.
18. Ferguson PC, McLaughlin CE, Griffin AM, et al. Clinical and functional outcomesof patients with a pathologic fracture in high-grade osteosarcoma. J SurgOncol2010; 102:120–124.
19. Yin K, Liao Q, Zhong D, Ding J, Niu B, Long Q, Ding D. Meta-analysis of limb salvage versusamputation for treating high-grade and localized osteosarcoma in patients with pathological fracture. ExpTher Med. 2012 Nov; 4(5):889-894.
20. Scully SP, Temple HT, O’Keefe RJ, et al. The surgical treatment of patients with osteosarcoma who sustain a pathological fracture. ClinOrthop. 1996; 324:227-232.
(Abstract Full Text HTML) (Download PDF)
Like this:
Biological Methods of Reconstruction After Excision of Extremity Osteosarcoma
Volume 2 | Issue 2 | May-Aug 2016 | Page 5-9 | Suman Byregowda, Ajay Puri, Ashish Gulia.
Authors: Suman Byregowda [1], Ajay Puri [1], Ashish Gulia [1].
1Orthopedic Oncology Services, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Address of Correspondence
Dr. Ashish Gulia
Associate Professor, Orthopedic oncology, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Email: aashishgulia@gmail.com
Abstract
The overall survival rates for non-metastatic osteosarcomas have dramatically improved from a mere 15-20% to 60-65% today. This was possible due a multifactorial improvement in all the disciplines and specifically the advent of multiagent chemotherapy. With an exponential increase in the survival as well as limb salvage procedures, it would be customary to invent cost effective, stable, durable reconstruction options. Various biological and non biological methods are available for reconstruction. In the era of metal and with the advent of growing artificial bones, non biological options appear to be an attractive and easily available option with excellent immediate results but their long term results and complications are debatable. On the other hand the less attractive biological methods are known to provide stable, durable, cost effective reconstruction options. In the present article we discuss various biological reconstruction methods available for extremity osteosarcoma patients, their advantages and disadvantages.
Keywords: Biological reconstruction , Osteogenic sarcoma
Introduction:
The era when osteosarcomas of the extremity were treated with only amputations is long past and the advent of multimodality management has completely changed the outcomes of these tumors. With newer chemotherapic agents, modern surgical techniques, better imaging techniques and affordable reconstructive options limb salvage has become the norm resulting in better functional and psychological outcomes The prerequisites for limb salvage include the ability to achieve an oncologically safe margin and ability to reconstruct the limb such that it provide better function compared to an amputation. Today this is possible in more than 95% of the patients [1]. Adequate oncologic clearance is paramount and the chosen method of reconstruction should never compromise the amount of resection required. The barriers to limb salvage are encasement of a major motor nerve, major vascular involvement, poorly placed biopsy incisions, uncontrolled infection, displaced pathological fractures and inadequate motors after resection of tumors. Besides fulfilling the basic pre requisites of limb salvage mentioned above the reconstructive modality chosen should permit an early return to daily activities and be aesthetically acceptable. . The reconstruction must be durable, economically feasible and should have minimum short term and long term complications. A number of reconstructions methods, both biological and non biological are available for the reconstruction of these skeletal defects after resection. The chosen method of reconstruction should be tailored for the individual based on the growth potential, site and amount of resection and functional requirements. This article discusses the biological techniques available for reconstruction of bone defects after resection of an extremity osteosarcoma.
Biological methods available for reconstructions are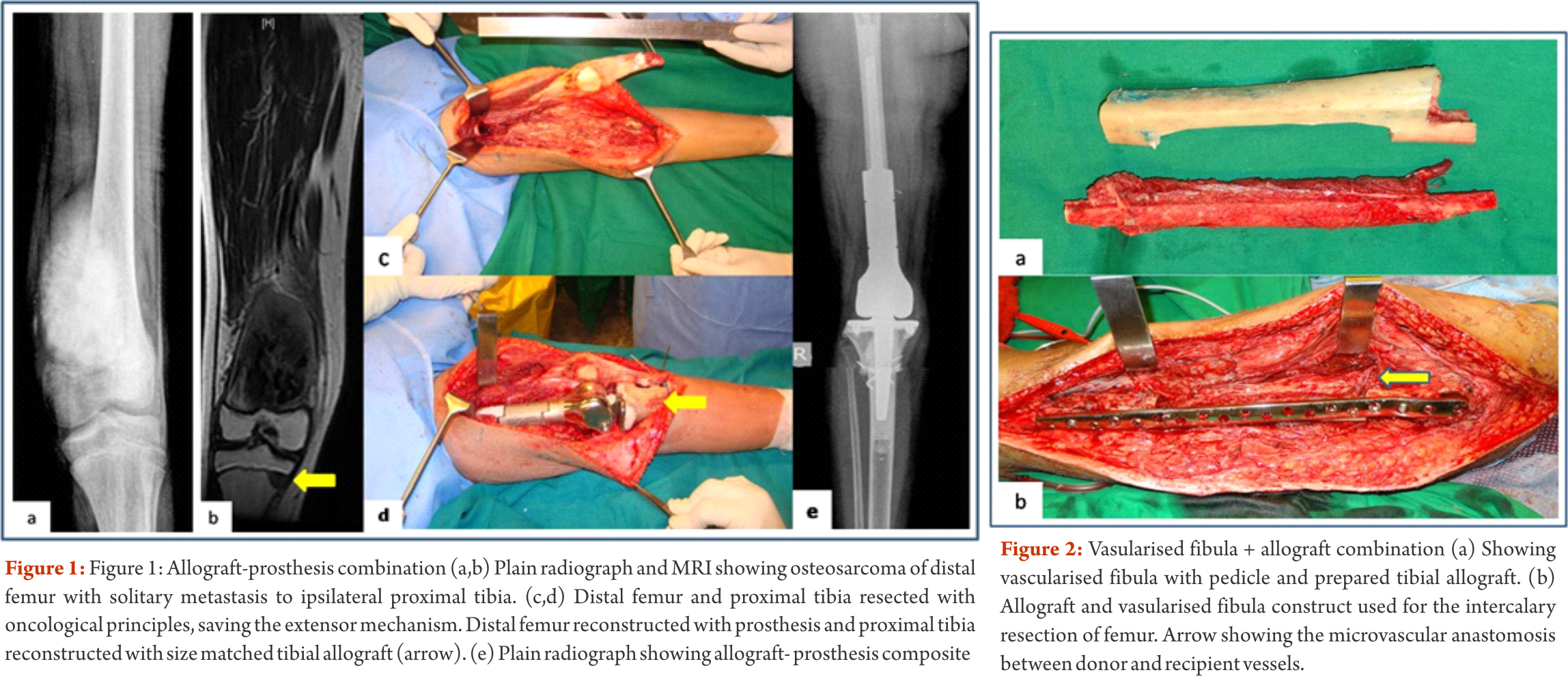 The functional outcomes with composite reconstruction are comparable with prosthetic reconstruction alone but associated with higher complication like nonunion and fracture. This method can have limited use in selected young patients with expected long term survival and require good bone stock for revision surgeries [4,5,6]. Allografts require sophisticated bone banks for procurement and storage and these are not available in most of the developing countries. Bone donations are not as frequent as other organ donations making procuring of size matched allografts even more challenging. Allografts may also be associated with risk of transmission of disease. Though strut allografts alone can be used for the reconstruction of intercalary defects and knee arthrodesis but studies have shown higher rate of complication like fracture, non union and resorption of grafts. Study by Bus MP et al has demonstrated a complication rate of 76 % and 70 % chance for reoperation due to graft failure. Thus strut allografts alone have limited use and are generally preferred for the upper limb or small defects (< 15cms). To overcome the above complication strut allograft may be combined with vascular fibular grafts [7,8]. Fibula is the most widely used autograft for reconstruction. It can be used as a vascularised or non vascularised graft. Proximal fibular head (with articular surface) has been used to reconstruct the articular surface of proximal humerus and distal radius. While isolated vascularized fibula may be adequate for reconstruction of upper limb defects where weight bearing is not an issue, lower limb reconstructions involving femur or the knee generally require a combination of vascularised fibula with strut allografts (Fig 2). Isolated use of fibula autograft or strut allografts have higher failure rates in large lower limb bone defects [9,10,11]. Small osteoarticular defects (up to 5 cm) like after the resection of distal radius lesions can also be reconstructed with iliac crest autograft. Certain anatomical sites have an inherent advantage and ease for reconstruction. Use of the neighbouring bone in forearm and leg provides a vascularised graft after resection of the radius and tibia. This serves as an easy and effective method of reconstruction. Shifting the distal ulna after an osteotomy at an appropriate level into the defect along with its soft tissue attachment and stabilizing it to the radius proximally and carpal bone distally (wrist arthordesis) provides an excellent method of reconstructing the bone defects after resection of distal radius tumors (Fig. 3). This method provides a stable wrist while maintaining forearm rotations (pronation- supination)[12]. Similarly in tibial lesions the fibula is mobilized medially into tibial defect and stabilized. This can be done both, for intercalary resections of the tibia where fibula is shifted after a double osteotomy and distal intrarticular resections where the transposed fibula is stabilized to talus to create an ankle arthrodesis This procedures avoids the requirement of a complex micro vascular procedure, reduces the operative time and also facilitates ease of soft tissue closure as transportation of fellow bone in to the defect will result in volume reduction of the tissues [13]. Reimplanting sterilized tumor host bone is widely used after intercalary resections. Patients own resected bone is sterilized and used to fill the defect. The resected tumor grafts can be sterilized by various methods like radiotherapy (extra-corporeal radiotherapy), pasteurization, autoclaving, liquid nitrogen and microwave. This technique has various advantages over the use of allograft. It does not require a bone bank, provides size matched graft (as it has been taken from the same defect) and has no risk of transmitted disease. After resection of the tumor the tumor bearing bone is taken on a separate table and soft tissues are removed under aseptic precautions. Certain soft tissues like ligaments may be retained on the bone graft in order to facilitate reconstruction. Sterilized bones are implanted back in the defect and stabilized with intramedullary nails or plates (Fig. 4).
The functional outcomes with composite reconstruction are comparable with prosthetic reconstruction alone but associated with higher complication like nonunion and fracture. This method can have limited use in selected young patients with expected long term survival and require good bone stock for revision surgeries [4,5,6]. Allografts require sophisticated bone banks for procurement and storage and these are not available in most of the developing countries. Bone donations are not as frequent as other organ donations making procuring of size matched allografts even more challenging. Allografts may also be associated with risk of transmission of disease. Though strut allografts alone can be used for the reconstruction of intercalary defects and knee arthrodesis but studies have shown higher rate of complication like fracture, non union and resorption of grafts. Study by Bus MP et al has demonstrated a complication rate of 76 % and 70 % chance for reoperation due to graft failure. Thus strut allografts alone have limited use and are generally preferred for the upper limb or small defects (< 15cms). To overcome the above complication strut allograft may be combined with vascular fibular grafts [7,8]. Fibula is the most widely used autograft for reconstruction. It can be used as a vascularised or non vascularised graft. Proximal fibular head (with articular surface) has been used to reconstruct the articular surface of proximal humerus and distal radius. While isolated vascularized fibula may be adequate for reconstruction of upper limb defects where weight bearing is not an issue, lower limb reconstructions involving femur or the knee generally require a combination of vascularised fibula with strut allografts (Fig 2). Isolated use of fibula autograft or strut allografts have higher failure rates in large lower limb bone defects [9,10,11]. Small osteoarticular defects (up to 5 cm) like after the resection of distal radius lesions can also be reconstructed with iliac crest autograft. Certain anatomical sites have an inherent advantage and ease for reconstruction. Use of the neighbouring bone in forearm and leg provides a vascularised graft after resection of the radius and tibia. This serves as an easy and effective method of reconstruction. Shifting the distal ulna after an osteotomy at an appropriate level into the defect along with its soft tissue attachment and stabilizing it to the radius proximally and carpal bone distally (wrist arthordesis) provides an excellent method of reconstructing the bone defects after resection of distal radius tumors (Fig. 3). This method provides a stable wrist while maintaining forearm rotations (pronation- supination)[12]. Similarly in tibial lesions the fibula is mobilized medially into tibial defect and stabilized. This can be done both, for intercalary resections of the tibia where fibula is shifted after a double osteotomy and distal intrarticular resections where the transposed fibula is stabilized to talus to create an ankle arthrodesis This procedures avoids the requirement of a complex micro vascular procedure, reduces the operative time and also facilitates ease of soft tissue closure as transportation of fellow bone in to the defect will result in volume reduction of the tissues [13]. Reimplanting sterilized tumor host bone is widely used after intercalary resections. Patients own resected bone is sterilized and used to fill the defect. The resected tumor grafts can be sterilized by various methods like radiotherapy (extra-corporeal radiotherapy), pasteurization, autoclaving, liquid nitrogen and microwave. This technique has various advantages over the use of allograft. It does not require a bone bank, provides size matched graft (as it has been taken from the same defect) and has no risk of transmitted disease. After resection of the tumor the tumor bearing bone is taken on a separate table and soft tissues are removed under aseptic precautions. Certain soft tissues like ligaments may be retained on the bone graft in order to facilitate reconstruction. Sterilized bones are implanted back in the defect and stabilized with intramedullary nails or plates (Fig. 4).  Reimplanted bone acts a scaffold for creeping substitution and incorporation. To enhance incorporation and the union at osteotomy sites they can be combined with a vasclarised fibula ( Capanna technique). Puri et al documented a mean union time of 7 months for osteotomy sites and an excellent MSTS score of 29 with extracorporeal radiotherapy [14,15]. To overcome the adverse events like nonunion, fracture and collapse with the use of liquid nitrogen to sterlise tumor bone (fresh frozen autograft), pedical autograft technique was developed. In this technique an osteotomy is done at one end or joint disarticulation done and the whole specimen is treated with liquid nitrogen with other end in continuity with the main bone. It is then stabilized back with internal fixation or athroplasty. As bony continuity is maintained at one end, it is presumed to have early blood flow recovery and faster union and less complication compared with frozen autograft [16,17]. The main drawback of sterilized bones are inadequate mechanical strength resulting in graft fracture and implant failure. To enhance incorporation and to overcome inadequate mechanical strength they can be combined with a vasclarised fibula ( Capanna technique). For surface lesions like periosteal, parosteal and high grade surface osteosarcomas where medullary canal is not involved, bone preserving hemicortical excision may be considered. Meticulous planning with MRI and CT scans are required to obtain adequate margins and preserve good native bone. Computer assisted navigation surgery is advantageous while performing such technically demanding bone preserving surgeries. Various options are available to fill the bone defect after hemicortical excision (sterlised resected bone, strut allografts or small defects can be filled with autografts) [18,19]. Rotationplasty involves converting ankle joint to knee joint by segmental resection and rotating the foot externally to 180 degrees. This is an alternative method for reconstruction especially in children with growth potential where cost constraints may preclude the use of expensive growing prosthesis. This worthies also useful in converting hind quarter or above knee amputations to a functional below knee like amputation in adult patients where conventional resection and reconstructions are not possible due to large or previously inappropriately treated lesions In distal femur and proximal tibia lesions, a segment of involved bone along with knee joint and involved soft tissues is removed only sparing the neurovascular bundle. Here the two segments are connected only with neurovascular bundle. The distal fragment is externally rotated 180 degrees and distal part of femur is stabilized to proximal tibia with appropriate implants, in such a way that the ankle comes to the level of opposite knee joint. In the cases with involvement of whole femur, proximal tibia is articulated with the hip joint with or without use of prosthesis after external 180 dgree rotation. Adequate soft tissue reconstructions and an intense rehabilitation protocol ensures an excellent functional outcome in these cases where the ankle will act like knee joint, dorsiflexion of ankle acts as flexion and plantar flexion acts as extension of knee joint (Fig. 5).
Reimplanted bone acts a scaffold for creeping substitution and incorporation. To enhance incorporation and the union at osteotomy sites they can be combined with a vasclarised fibula ( Capanna technique). Puri et al documented a mean union time of 7 months for osteotomy sites and an excellent MSTS score of 29 with extracorporeal radiotherapy [14,15]. To overcome the adverse events like nonunion, fracture and collapse with the use of liquid nitrogen to sterlise tumor bone (fresh frozen autograft), pedical autograft technique was developed. In this technique an osteotomy is done at one end or joint disarticulation done and the whole specimen is treated with liquid nitrogen with other end in continuity with the main bone. It is then stabilized back with internal fixation or athroplasty. As bony continuity is maintained at one end, it is presumed to have early blood flow recovery and faster union and less complication compared with frozen autograft [16,17]. The main drawback of sterilized bones are inadequate mechanical strength resulting in graft fracture and implant failure. To enhance incorporation and to overcome inadequate mechanical strength they can be combined with a vasclarised fibula ( Capanna technique). For surface lesions like periosteal, parosteal and high grade surface osteosarcomas where medullary canal is not involved, bone preserving hemicortical excision may be considered. Meticulous planning with MRI and CT scans are required to obtain adequate margins and preserve good native bone. Computer assisted navigation surgery is advantageous while performing such technically demanding bone preserving surgeries. Various options are available to fill the bone defect after hemicortical excision (sterlised resected bone, strut allografts or small defects can be filled with autografts) [18,19]. Rotationplasty involves converting ankle joint to knee joint by segmental resection and rotating the foot externally to 180 degrees. This is an alternative method for reconstruction especially in children with growth potential where cost constraints may preclude the use of expensive growing prosthesis. This worthies also useful in converting hind quarter or above knee amputations to a functional below knee like amputation in adult patients where conventional resection and reconstructions are not possible due to large or previously inappropriately treated lesions In distal femur and proximal tibia lesions, a segment of involved bone along with knee joint and involved soft tissues is removed only sparing the neurovascular bundle. Here the two segments are connected only with neurovascular bundle. The distal fragment is externally rotated 180 degrees and distal part of femur is stabilized to proximal tibia with appropriate implants, in such a way that the ankle comes to the level of opposite knee joint. In the cases with involvement of whole femur, proximal tibia is articulated with the hip joint with or without use of prosthesis after external 180 dgree rotation. Adequate soft tissue reconstructions and an intense rehabilitation protocol ensures an excellent functional outcome in these cases where the ankle will act like knee joint, dorsiflexion of ankle acts as flexion and plantar flexion acts as extension of knee joint (Fig. 5). 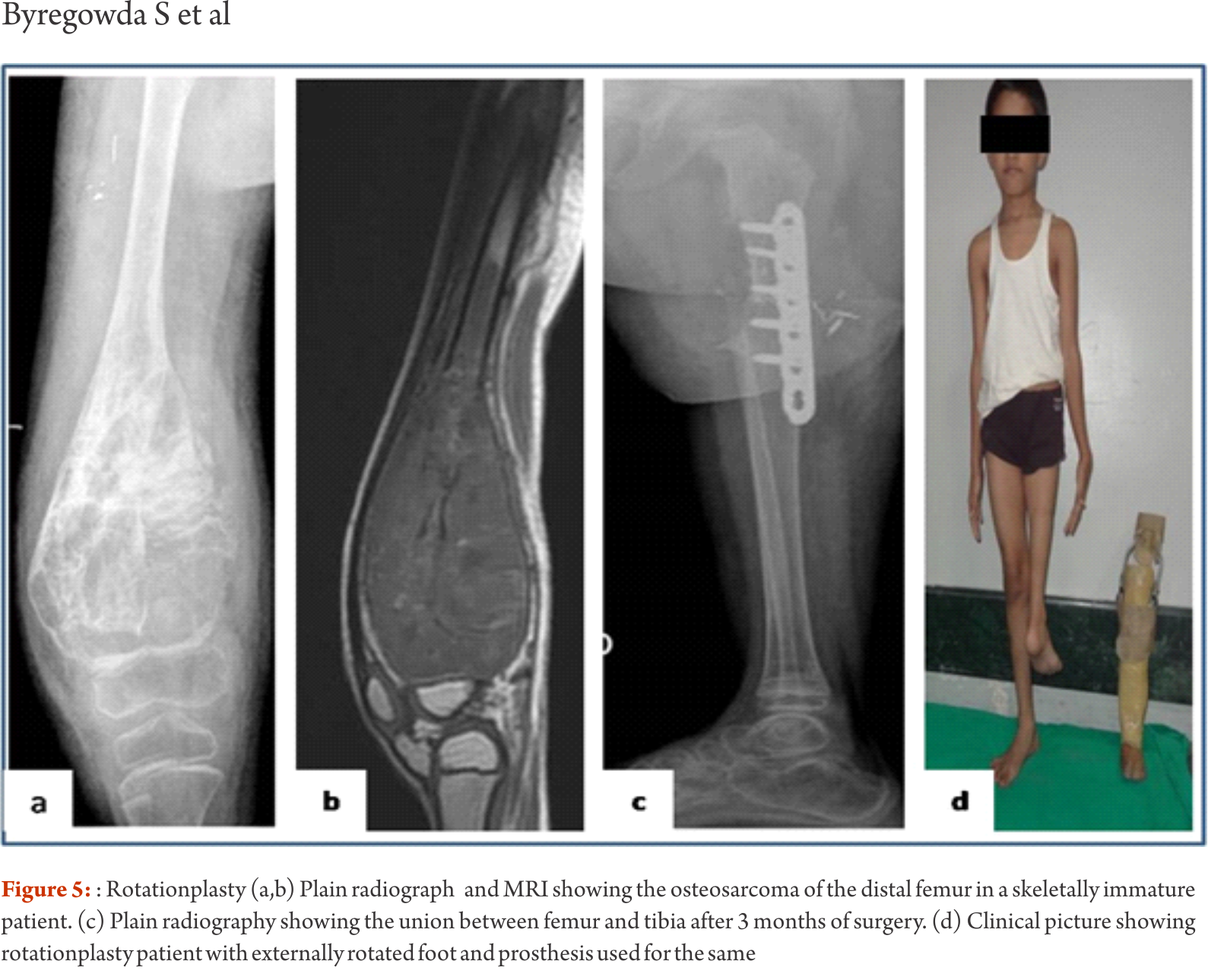 This procedure can also be used as salvage surgery following infected and failed limb salvage reconstruction. Studies have shown excellent oncological and functional outcome with this procedure. Rotationplasty offers a durable reconstruction option. It is not associated with phantom limb pain or sensations which are common following amputations. The main drawback of the procedure is the cosmetic deformity due to posterior rotated foot [20,21]. Distraction osteogensis using ilizarov method has been used for bone defects in tumor resection. It can be combined with live fibula grafts. The disadvantages are prolonged duration of treatment, high incidence of pin tract infections due to immune compromised state of patient receiving chemotherapeutic agents [22,23]. Due to these complications it is not a popular method and is used rarely. Masquelet technique is a two stage procedure for reconstruction of bone defects. In the first stage the defect is filled with bone cement and stabilized. This leads to the formation of a biological membrane over the cement spacer. In second stage procedure the biological membrane is opened, cement spacer removed, filled with cortico-cancellous bone graft and biological membrane sutured to create close content. The procedure was described in children. The ideal time for stage two is between 6 to 8 weeks, though in oncology we wait for completion of adjuvant treatment. Advantage is it makes primary surgery short and rapid uptake of graft due to biological membrane after the second procedure. The disadvantage with procedure is requirement of two surgical interventions [24].
This procedure can also be used as salvage surgery following infected and failed limb salvage reconstruction. Studies have shown excellent oncological and functional outcome with this procedure. Rotationplasty offers a durable reconstruction option. It is not associated with phantom limb pain or sensations which are common following amputations. The main drawback of the procedure is the cosmetic deformity due to posterior rotated foot [20,21]. Distraction osteogensis using ilizarov method has been used for bone defects in tumor resection. It can be combined with live fibula grafts. The disadvantages are prolonged duration of treatment, high incidence of pin tract infections due to immune compromised state of patient receiving chemotherapeutic agents [22,23]. Due to these complications it is not a popular method and is used rarely. Masquelet technique is a two stage procedure for reconstruction of bone defects. In the first stage the defect is filled with bone cement and stabilized. This leads to the formation of a biological membrane over the cement spacer. In second stage procedure the biological membrane is opened, cement spacer removed, filled with cortico-cancellous bone graft and biological membrane sutured to create close content. The procedure was described in children. The ideal time for stage two is between 6 to 8 weeks, though in oncology we wait for completion of adjuvant treatment. Advantage is it makes primary surgery short and rapid uptake of graft due to biological membrane after the second procedure. The disadvantage with procedure is requirement of two surgical interventions [24].
A) Allografts
B) Autografts – vascularised and non vascularised
C) Patient’s own sterilized tumor bone
D) Combination of allografts/ sterilized tumor bone and vascularised autografts
E) Distraction osteogenesis with Ilizarov technique
F) Rotationplasty
G) Masquelet technique
Depending on the extent of the resection, the surgical resections can be categorised as Osteo-articular resections and Intercalary resections. Reconstruction after osteoarticular resections is mainly done by megaprosthesis (non biological). If you want to retain joint mobility the biological options available are limited to osteoarticular allografts. Though these maintain bone stock and provide a better attachment for surrounding soft tissue resulting in increased stability of the construct the long term results with osteoarticular allografts are disappointing .Fracture, arthritis, non unions, infections and repeated surgery are not uncommon. Studies have reported 60-70 % adverse events, overall 5 year survival of 69 % and 79 % for allograft and articulate surface respectively[2,3]. A composite of allograft and prosthesis has been widely used, where allograft helps to maintain the stock and prosthesis provides the articular surface (Fig. 1).
Conclusions
Reconstruction following tumor resection is a challenging task. Different biological and non biological methods are available. Selection of a reconstruction procedure should be tailored to the individual patient based on the bone affected, amount of resection, requirement of patient and expertise and infrastructure available at treating centre. Biological methods are more cost effective and provide durable reconstruction options in properly selected extremity osteosarcoma patients.
References
1. Ghert M, Deheshi B, Holt G, Randall RL, Ferguson P, Wunder J et al.Prophylactic antibiotic regimensin tumour surgery (PARITY): protocol for a multicentre randomised controlledstudy. BMJ Open. 2012 Nov 28;2(6).
2. Muscolo DL, Ayerza MA, Farfalli G, Aponte-Tinao LA. Proximal tibia osteoarticular allografts in tumor limb salvage surgery. Clin OrthopRelat Res. 2010 May;468(5):1396-404.
3. Toy PC, White JR, Scarborough MT, Enneking WF, Gibbs CP. Distal femoral osteoarticular allografts: long-term survival, but frequent complications. Clin OrthopRelat Res. 2010 Nov;468(11):2914-23.
4. Donati D, Colangeli M, Colangeli S, Di Bella C, Mercuri M. Allograft-prosthetic composite in the proximal tibia after bone tumor resection. Clin OrthopRelat Res. 2008;466:459–65.
5. Van de Sande MA, Dijkstra PD, Taminiau AH. Proximal humerus reconstruction after tumour resection:Biological versus endoprosthetic reconstruction. IntOrthop. 2011;35:1375–80.
6. Campanacci L, Manfrini M, Colangeli M, Alí N, Mercuri M. Long-term results in children with massive bone osteoarticular allografts of the knee for high-grade osteosarcoma. J PediatrOrthop. 2010 Dec;30(8):919-27.
7. Bus MP, Dijkstra PD, van de Sande MA, Taminiau AH, Schreuder HW, Jutte PC, van der Geest IC et al. Intercalary allograft reconstructions following resection of primary bone tumors: a nationwide multicenter study. J Bone Joint Surg Am. 2014 Feb 19;96(4):e26.
8. Aponte-Tinao LA, Ayerza MA, Muscolo DL, Farfalli GL. Allograft reconstruction for the treatment of musculoskeletal tumors of the upper extremity. Sarcoma.2013;2013:925413.
9. Scaglioni MF, Chang EI, Gur E, Barnea Y, Meller I, Kollander Y, Bickels J, Dadia S, Zaretski A. The role of the fibula head flap for joint reconstruction after osteoarticular resections. J PlastReconstrAesthet Surg. 2014 May;67(5):617-23.
10. Campanacci DA, Puccini S, Caff G, Beltrami G, Piccioli A, Innocenti M, Capanna R. Vascularised fibular grafts as a salvage procedure in failed intercalary reconstructions after bone tumour resection of the femur. Injury. 2014 Feb;45(2):399-404.
11. Hilven PH, Bayliss L, Cosker T, Dijkstra PD, Jutte PC, Lahoda LU, Schaap GR, Bramer JA, van Drunen GK, Strackee SD, van Vooren J, Gibbons M, Giele H, van de Sande MA. The vascularised fibular graft for limb salvage after bone tumour surgery: a multicentre study. Bone Joint J. 2015 Jun;97-B(6):853-61.
12. Puri A, Gulia A, Agarwal MG, Reddy K. Ulnar translocation after excision of a Campanacci grade-3 giant-cell tumour of the distal radius: an effective method of reconstruction. J Bone Joint Surg Br. 2010 Jun;92(6):875-9.
13. Puri A, Subin BS, Agarwal MG. Fibular centralisation for the reconstruction of defects of the tibial diaphysis and distal metaphysis after excision of bone tumours. J Bone Joint Surg Br. 2009 Feb;91(2):234-9.
14. Mottard S, Grimer RJ, Abudu A, Carter SR, Tillman RM, Jeys L, Spooner D. Biological reconstruction after excision, irradiation and reimplantation of diaphyseal tibial tumours using an ipsilateralvascularised fibular graft. J Bone Joint Surg Br. 2012 Sep;94(9):1282-7.
15. Puri A, Gulia A, Jambhekar N, Laskar S. The outcome of the treatment of diaphyseal primary bone sarcoma by resection, irradiation and re-implantation of the host bone: extracorporeal irradiation as an option for reconstruction in diaphyseal bone sarcomas. J Bone Joint Surg Br. 2012 Jul;94(7):982-8.
16. Igarashi K, Yamamoto N, Shirai T, Hayashi K, Nishida H, Kimura H, Takeuchi A, Tsuchiya H. The long-term outcome following the use of frozen autograft treated with liquid nitrogen in the management of bone and soft-tissue sarcomas. Bone Joint J. 2014 Apr;96-B(4):555-61.
17. Shimozaki S, Yamamoto N, Shirai T, Nishida H, Hayashi K, Tanzawa Y, Kimura H, Takeuchi A, Igarashi K, Inatani H, Kato T, Tsuchiya H. Pedicle versus free frozen autograft for reconstruction in malignant bone and soft tissue tumors of the lower extremities. J Orthop Sci. 2014 Jan;19(1):156-63.
18. Deijkers RL, Bloem RM, Hogendoorn PC, Verlaan JJ, Kroon HM, TaminiauAH.Hemicortical allograft reconstruction after resection of low-grade malignant bone tumours. J Bone Joint Surg Br. 2002 Sep;84(7):1009-14.
19. Agarwal M, Puri A, Anchan C, Shah M, Jambhekar N. Hemicortical excision for low-grade selected surface sarcomas of bone. Clin OrthopRelat Res. 2007 Jun;459:161-6.
20. Agarwal M, Puri A, Anchan C, Shah M, Jambhekar N. Rotationplasty for bone tumors: is there still a role? Clin OrthopRelat Res. 2007 Jun;459:76-81.
21. Gradl G, Postl LK, Lenze U, Stolberg-Stolberg J, Pohlig F, Rechl H, Schmitt-Sody M, von Eisenhart-Rothe R, Kirchhoff C. Long-term functional outcome and quality of life following rotationplasty for treatment of malignant tumors. BMC MusculoskeletDisord. 2015 Sep 24;16:262.
22. Demiralp B, Ege T, Kose O, Yurttas Y, Basbozkurt M. Reconstruction of intercalary bone defects following bone tumor resection with segmental bone transport using an Ilizarov circular external fixator. J Orthop Sci. 2014 Nov;19(6):1004-11.
23. Khira YM, Badawy HA. Pedicled vascularized fibular graft with Ilizarov external fixator for reconstructing a large bone defect of the tibia after tumor resection. J OrthopTraumatol. 2013 Jun;14(2):91-100.
24. Chotel F, Nguiabanda L, Braillon P, Kohler R, Bérard J, Abelin-Genevois K. Induced membrane technique for reconstruction after bone tumor resection in children: a preliminary study. OrthopTraumatolSurg Res. 2012 May;98(3):301-8.
(Abstract Full Text HTML) (Download PDF)
Like this:
Tata Memorial Centre – The Journey so Far!!!
Volume 2 | Issue 2 |May- Aug 2016 | Page 3-4 | Ashish Gulia, Ajay Puri, Rajendra.A. Badwe
Authors: Ashish Gulia [1], Ajay Puri [1], Rajendra.A. Badwe [1].
[1]Orthopedic Oncology Services, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Address of Correspondence
Dr. Ashish Gulia
Associate Professor, Orthopedic oncology, Department of Surgical Oncology, Tata Memorial Hospital, Mumbai.
Email: aashishgulia@gmail.com
Tata Memorial centre, considered as “Mecca” of Oncology care in the country and sub continent, stands today at its Dodranscentennial Anniversary. Institution started as Tata Memorial Hospital, which was inaugurated on 28th February 1941 by His Excellency Sir Roger Lumley, then Governor of Mumbai. Declaring the centre open, he estimated with extraordinary prescience “…the hospital will become the spearhead of attack on cancer in this country, providing not only a center where specialized treatment can be given, but also one from which knowledge of new methods of treatment and diagnosis will go out to doctors and hospitals throughout the country…”The past 75 years stand as glowing testimony to Sir Lumley’s prophecy. A recent peer group review of the hospital has accredited it as the premier comprehensive center in India, in addition recognized it as potentially and prominently featuring in the top five cancer centers globally. The advent of TMH in chapters of the history of medicine in India can best be defined as a quantum leap. It owed its genesis to the philanthropic sentiments of the illustrious Tata family. Later on, in 1961 was passed under the guardianship of Department of Atomic Energy which remarkably aided and upheld the meteoric rise of the sterling institution. In the year 1966 Tata Memorial Hospital merged with Cancer Research Institute (the pioneer research institute), the conjoined institutes were collectively called Tata Memorial Centre (TMC). TMC exemplifies private philanthropy boosted by Government support with a mandate for Service, Education & Research in Cancer. Commencing its journey as a torchbearer, the first dedicated cancer specialty hospital in Asia, TMC today continues to maintain its excellence of being the largest cancer institute in Asia. A fledgling 80 bedded hospital spread over 15,000 sq meters with an annual budget of half a million (INR), has escalated to a lofty institution with 700+ beds, covering over 75,000 sq meters, utilizing an annual budget of 23,00 million (INR). TMC indomitably bears the responsibility of cancer care burden not only of the large Indian populace but also several parts of Asia, Middle East and Africa, catering to an annual footfall of 65,000 new cases and 450,000 follow up cases.[1] Research and education have unarguably been the essential arms of a comprehensive cancer care centre. TMC since origin has embraced the holistic model of delivering cancer care by augmenting and developing its research and education facilities as it sought to provide treatment which was affordable, innovative and particularly relevant to the needs of the country. The research wing of TMC originally established in 1952 as CRI (Cancer Research Institute) was subsequently revamped as ACTREC (Advanced Centre for Treatment Research and Education in Cancer) in 2002. ACTREC is a state of art research and training centre which has over the years focussed on the integration of basic and clinical research including evolving inspiring pathways for translational research. The academic unit of TMC imparts training in specialty and super specialty courses in oncology and is affiliated to the acclaimed Homi Bhabha National Institute, Mumbai.[1]
The noble mission of the institute was to provide comprehensive cancer care to one and all. A practice which has been resolutely followed since its inception is, that every patient who walks in is attended to and treated irrespective of their ability to pay. Over 60% of TMC’s patients receive free or highly subsidized treatment. The essence of the center however, lies in its undeviating constancy towards extending compassionate care and assuaging suffering in times of extreme distress. Patients and caregivers often refer to the hospital as a temple which bespeaks diligence and dedication of the individuals who work there. As millions stream in seeking hope, this Mecca continually labors to ensure means of cure, care and cope. Seventy five glorious years of Tata Memorial Centre have borne witness to its commitment to excellence, expertise and eminence in cancer treatment, care, research, education and awareness. As a tribute to this iconic institution we mark a yearlong celebration of its Platinum Jubilee (Dodranscentennial Anniversary) 2016.[1] The appointment of a full time orthopaedic surgeon on the staff of Tata Memorial Centre in 1999 resulted in the establishment of a specialist orthopaedic oncology service [2]. Over the years this service has now grown to be recognised as one of the leading musculoskeletal oncology units globally. Like all other services at the centre this too functions as a “Disease Management Group”, where specialists from all disciplines involved in sarcoma care (radiology, pathology, medical oncology, radiation oncology, etc.) come together to provide multidisciplinary integrated care under one umbrella. Today the orthopaedic oncology service caters to about 2500 new musculoskeletal oncology cases per year while performing about 700 major and 1200 minor surgical procedures each year. The service has collaborated with industry to develop cost effective mechanical reconstructive options suited to the local socio economic milieu. The development of an indigenous prosthesis (TMH – NICE / RESTOR) which was recognised by the Golden Peacock award for health innovation is a proud feather in the cap of the institute. Innovative biological reconstruction techniques published in reputed peer reviewed journals are now practised in other centres globally too. Both clinical and basic research also plays a part in the units’ contribution to furthering insights in sarcoma care. The service has to its credit the only randomised trial ever conducted for surveillance in sarcomas and is part of a global consortium that has published one of the largest series of patients detailing the genomic profile of sarcomas. [3,4] Imparting training and sharing of knowledge and information has always been part of the culture of this institute and a host of young orthopaedic oncologists practising all across the country are a proud testimony in keeping with this tradition. There is a constant stream of international trainees and visitors and the service has helped in initiating and developing the limb salvage program in many resource challenged areas like Nepal, Myanmar, Palestine and Nigeria. [5] The TMC “Evidence Based Guidelines” for the management of bone and soft tissue sarcomas is a useful handbook that helps disseminate the concept of scientific and rational treatment for these challenging tumors. TMC staff and alumni have been at the forefront in establishing the Indian Musculo Skeletal Oncology Society (IMSOS), an organisation that seeks to provide a common forum for interaction and mutual collaboration between different specialists and institutes involved in the treatment of sarcomas. As the institute seeks to expand its footprint over the next decade by adding infrastructure and staff, both in Mumbai and centres across the country the orthopaedic oncology service too will need to evolve. Challenges of adopting new technology will have to be met. Yet, offering the right balance between these enhanced options while maintaining the principles of rational , cost effective medical care will necessitate “the wisdom of Solomon”.
Just as it has for the past 75 years, the institute and every individual connected with it will always strive with utmost passion to fulfil its motto of “Service – Education – Research” in a ceaseless endeavour to offer the best possible care to the patients that repose their trust in this institute.
References
1. Tata Memorial Centre. 2016. Tata Memorial Centre Platinum Jubilee – Celebrating 75 Glorious Years. Available at: http://tmcplatinumjubilee.org/tmc-platinum-jubilee.php
2. Puri A – The “ODYSSEY”: “Orthopaedic Oncology” – My journey thus far! Journal of Bone and Soft Tissue Tumors May-Aug 2015; 1(1):4-6
3. Puri A, Gulia A, Hawaldar R, Ranganathan P, Badwe RA. Does Intensity of Surveillance Affect Survival After Surgery for Sarcomas? Results of a Randomized Noninferiority Trial. ClinOrthopRelat Res. 2014 May;472(5):1568-75.
4. Ballinger ML, Goode DL, Ray-Coquard I, James PA, Mitchell G, Niedermayr E, Puri A, Schiffman JD, Dite GS, Cipponi A, Maki RG, Brohl AS, MyklebostO,Stratford EW, Lorenz S, Ahn SM, Ahn JH, Kim JE, Shanley S, Beshay V, Randall RL, Judson I, Seddon B, Campbell IG, Young MA, Sarin R, Blay JY, O’DonoghueSI,Thomas DM; International Sarcoma Kindred Study. Monogenic and polygenic determinants of sarcoma risk: an international genetic study. Lancet Oncol. 2016 Aug 4.
5. Gulia A, Wilding C, Suman MB, Puri A. OrthOncoCon-2014: Continuing education in musculoskeletal oncology. Indian J Cancer. 2015 Apr-Jun;52(2):184-5.
(Abstract Full Text HTML) (Download PDF)
Like this:
Journal of Bone and Soft Tissue Tumors – Unique Status
Vol 2 | Issue 2 | May – Apr 2016 | page:1-2 | Dr Yogesh Panchwagh & Dr Ashok Shyam.
Author: Dr Yogesh Panchwagh [1] & Dr Ashok Shyam [1,2].
[1]Orthopaedic Oncology Clinic, Pune, India.
[2] Indian Orthopaedic Research Group, Thane, India
[3] Sancheti Institute for Orthopaedics &Rehabilitation, Pune, India.
Address of Correspondence
Dr. Yogesh Panchwagh.
Orthopaedic Oncology Clinic, 101, Vasant plot 29, Bharat Kunj Society -2, Erandwana, Pune – 38, India.
Email: drpanchwagh@gmail.com
Journal of Bone and Soft Tissue Tumor (JBJST) enjoys a special status in being one of the unique journals exclusively dedicated to bone and soft tissue tumors. The clinical expertise and technological development has been very rapid in this field and JBST already had a demand among the clinicians involved in taking care of musculoskeletal Oncology. JBST has successfully filled this vacuum that existed and has received great support from musculoskeletal tumor surgeons and clinicians. The platform provided by JBST has been used worldwide to access articles and also to submit original research. There are other factors that further add to its uniqueness. One of the most unique point is that this is a journal that is initiated by clinicians. JBST was conceived and initiated by people who were directly involved in care of musculoskeletal tumor patients. They perceived the need of such a journal and were instrumental in moulding it in its current shape. Another unique point of JBST is that it is not a pure research journal but also a tool to educate the young trainees and practitioners. JBST has a dedicated symposium in each issue which provides a comprehensive overview of the subject along with recent updates. This is very helpful for students, trainees and practitioners of the subject. These symposium articles are written by carefully solicited authors that have years of practical experience which add to the flavour of the article. The authors are requested to add the practical tips and cases to these symposium reviews to make it much more clinically relevant rather than simply publish a theoretical review of literature. Special attention is given to students in JBST and a students corner is published in every issue which is a brief overview article on a single bone tumor. This is specially created with keeping students in mind and is co-created with help of a trainee or a student. This has received excellent response as far as student readership is concerned and we thank Dr Ashish Gulia for initiating and supporting this initiative. Oncomedia is another unique part of JBST where recent conferences, upcoming conferences and new updates regarding bone and soft tissue tumors are listed. Videos and other academic materials are also included in this section. It’s a very unique source of information to our readers and is presented in a very reader friendly manner.This particular issue is also unique as it features and is dedicated to one of the most prestigious orthopaedic oncology unit in India from Tata Memorial Hospital (TMH) in Mumbai. The guest editorial is been contributed by the TMH team and it traces the journey of the hospital from its inception till today. JBST plans to continue this feature where such units, who have contributed significantly to growth of musculoskeletal oncology will be featured. We intend to include this a regular feature in JBST. Another feature that we really wish to include is academic interview, featuring individual personalities in the field of musculoskeletal oncology. Other journals from orthopaedic research group like trauma international and International Journal of Paediatric Orthopaedics have been regularly publishing such academic interviews. Hopefully JBJST will start this feature by next year. The JBST team is always looking for making the journal better with the aim to provide the best content to its reader which is presented in the most accessible and easy format. Being focussed on the subject of bone and soft tissue tumours and created and run by focussed and expert team has helped the journal achieve a unique status already and with help of the entire fraternity it is sure to grow further. If you have any suggestions for the editorial team, please feel free to write to us.
Dr Yogesh PachwaghDr Ashok Shyam
(Abstract Full Text HTML) (Download PDF)
Like this:
PVNS talus in a patient treated for chondral lesion in ipsilateral calcaneum: A case report and review of literature
Vol 3 | Issue 2 | Sep-Dec 2017 | Page 10-13 | Apurv Gabrani, Hitesh Dawar, Deepak Raina, Surbhit Rastogi
Authors: Apurv Gabrani [1], Hitesh Dawar [1], Deepak Raina [1], Surbhit Rastogi [1].
[1] Indian Spinal Injuries Centre, New Delhi.
Address of Correspondence
Dr . Apurv Gabrani
Ah-22, Shalimar Bagh, New Delhi-110088
Email: apurvgabrani@gmail.com
Abstract
Introduction: PVNS is a locally aggressive synovial proliferative disorder of unknown etiology and has been described in the foot and ankle in previous literature. A case of PVNS in the talus has been described in a patient treated for ipsilateral calcaneal chondral lesion.
Case Report: A 56 year old male presented with pain in his left ankle of 4 months duration. On investigation, he was found to have a well defined lytic lesion in the left calcaneum on x-ray. MRI showed a hyper intense lesion on T2WI. A needle biopsy revealed chondrogenic tumor which was managed by extended curettage. At 12 months follow up, patient presented with recent onset pain over the anterior aspect of left ankle which showed hypo density over the supero-anterior aspect of the talus and MRI showed ill defined hypo intense lesion on T2WI and hyper intense lesion on T1WI. The lesion increased in size on repeat MRI 6 weeks later. He was managed with synovectomy and debridement with core needle biopsy of talus. Histopathological examination revealed features consistent with PVNS. Patient remains asymptomatic at 1 year follow up after surgery.
Conclusion: A double primary lesion although rare, does exist and any recurrence should be viewed at with equal degree of suspicion as the primary lesion.
Keywords: Pigmented villonodular synovitis (PVNS), Talus, Calcaneum, Double Primary lesion, Chondral lesion.
References
1. Granowitz S.P., D’Antonio, j., and Mankin, H.L. The pathogenesis and long term results of PVNS. Clin Orthop 1976 ;114:335-351
2. Bakotic BW, Borkowski P. Primary soft tissue neoplasms of the foot: the clinicopathological features of 401 cases. J Foot Ankle Surg 2001; 40:29-36
3. T. Okoro, S. Isaac, R.U. Ashford, C.J. Kershaw. Pigmented villonodular synovitis of the talonavicular joint: A case report and review of the literature. The Foot 19(2009) 186-188
4. Jaffe HL, Litchtenstein L, Sutro C. Pigmented Villonodular Synovitis: bursitis and tenosynovitis. Arch Pathol 1941;31:731-65
5. Llauger J, Palmer J, Roson N. Pigmented villonodular synovitis and giant cell tumors of the tendon sheath: radiologic and pathologic features. Am J Roentgenol 1999; 172(4): 1087-91
6. Dorwart R.H., Genant H.K., Johnston W.H., and Morris J.M. PVNS of synovial joints: clinical, pathological, and radiologic features. Am J Roentgenol 1984;143:877-885
7. Sharma H, Jane MJ, Reid R. Pigmented villonodular synovitis of the foot and ankle: forty years of experience from the Scottish Bone Tumor Registry. J Foot Ankle Surg 2006;45(5): 329-36
8. Carmon W.A. Pigmented villonodular synovitis. Med Clin North Am 1947; 49:26-38
9. Young JM, Hudacek AG. Experimental production of pigmented villonodular synovitis in dogs. Am J pathol 1954;30:799
10. DeBruin J.A and Rockwood C.A. PVNS: Invasion of bone involving the knee joint. South Med J 1956; 59:466-468
11. McMaster P.E. PVNS with invasion of bone. J Bone Joint Surg 1960; 42A:1170-1183
12. Rothstein A.S. Localized PVNS of the ankle. J Am Podiatr Med Assoc1981; 71:607-610
13. Chung S.M.D., Janes J.M. Diffuse PVNS of the hip joint. J Bone Joint Surg1965; 47A:293-303
14. Bravo S.M., Winalski C.S., Weissman B.N. Pigmented villonodular synovitis. Radiol Clin North Am 1996; 34:311-26
15. Lin J, Jacobson JA, Jamadar DA, Ellis JH. Pigmented villonodular synovitis and related lesions: the spectrum of imaging findings. Am J Roentgenol 1999;172:191-7
16. Berger. I, Ehemann. V, Helmchen. B, Penzel. R, Weckauf. H. Comparative analysis of cell populations involved in the proliferative and inflammatory process in localized and diffuse pigmented villonodular synovitis: Histopathol 2004 Jul;19-3:687-92
17. Lewis R.W. Roentgen diagnosis of PVNS and synovial SA of the knee joint. Radiology1947; 49:26-38
18. Ho C.F. Chiou H.J., Chou Y.H. Peritendinous lesions. The role of high resolution ultrasonography. J. Clin. Imaging 2003;27:239-250
19. Ugai K., Morimoto K. Magnetic resonance imaging of pigmented villonodular synovitis in subtalar joint. Report of a case: Clin Orthop 1992;283:281-4
20. Myers B.W, Masi A.T. Pigmented villonodular synovitis and tenosynovitis: a clinical epidemiological study of 166 cases and literature review. Medicine(Baltimore)1980;59(3):223-38
21. Al-Nakshabandi N.A., Ryan A.G., Choudur H.: Pigmented villonodular synovitis. Clin Radiol 2004;59:414-20
22. Babinas I., De silva U., Grimer R.J. Pigmented villonodular synovitis of the foot and ankle: a 12 year experience from a tertiary orthopedic oncology unit. J Foot Ankle Surg. 2004;43(6):407-11
23. Segler C.P. Irradiation as an adjunctive treatment of diffuse pigmented villonodular synovitis of the foot and ankle prior to tumor surgical excision. Med Hypothesis 2003;61:229-230
24. Byers P.D., Cotton R.E., Deacon O.W., Lowy M., Newmau P.H., Sissons H.A., Thomson A.D.: The diagnosis and treatment of pigmented villonodular synovitis. J Bone Joint Surg1968; 50:290.
(Abstract Full Text HTML) (Download PDF)
Like this: